INTRODUCTION
The anterior choroidal artery (AchoA) supplies several important structures including the optic tract, geniculate body, posterior limb of internal capsule, globus pallidus, cerebral peduncle, uncus, amygdala, ventrolateral nucleus of thalamus, and choroid plexus. The vascular injury of AchoA may result in severe neurological deficits such as hemiplegia, hemianesthesia, hemianopsia, decreased consciousness, and extrapyramidal symptoms. Therefore, complete knowledge about vascular anatomy of AchoA is very important before surgical or endovascular manipulations. Although several detailed reports about anatomical variations of AchoA have been reported, transposition of AchoA as to posterior communicating artery (PcomA) origin was demonstrated only in 3 reports2-4). We report a case with ruptured cerebral aneurysm arising from PcomA, which originated from the internal carotid artery (ICA) distal to the origin of AchoA.
CASE REPORT
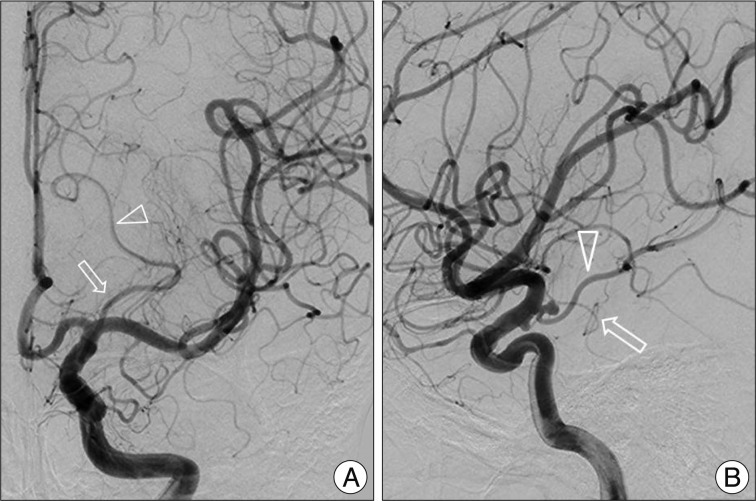
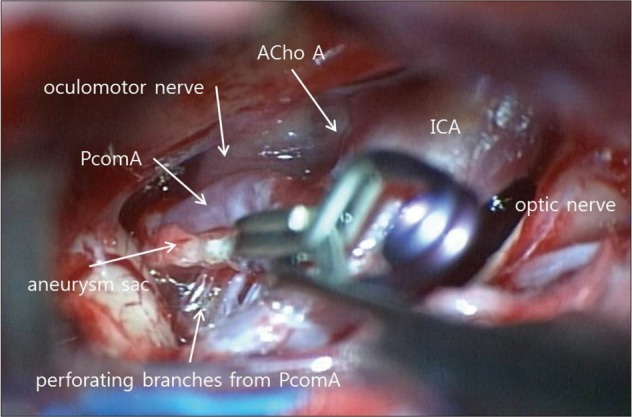
A 40-year-old woman presented with sudden onset of headache. Computed tomography (CT) revealed subarachnoid hemorrhage (SAH) which was slightly predominant in the left basal and sylvian cisterns. Two small aneurysms arose from both distal supraclinoid ICA, these were observed on CT angiograms (Fig. 1). Cerebral angiography demonstrated a small aneurysm arising from left fetal type of PcomA with suspicion of rupture and the other small aneurysm originating from right PcomA or perforating branch of ICA. In this study, left AChoA originated from ICA proximal to the origin of fetal type of PcomA (Fig. 2). The patient underwent neck clipping of left PcomA aneurysm. These findings were confirmed during operation (Fig. 3).
DISCUSSION
AchoA usually arises from the posterolateral wall of ICA, a few mm distal to the origin of PcomA, and it provides blood supply to the important neural structures around crural and ambient cisterns. These regions may also receive perforating branches arising from ICA frequently. The AchoA can have several variations in the course, size, number, branching pattern, and brain region supplied by this artery1). Among them, there have been several anomalies related to the origin of AchoA including transposition of AchoA and PcomA, origin from PcomA, origin from junction of ICA and PcomA, and origin from middle cerebral artery4,5). The clinical findings of transposed AchoA cases including present one are summarized in Table 1. All of the transpositions of AchoA occurred in the left side. Among all the cases, only two were associated with ipsilateral ruptured cerebral aneurysms arising from AchoA or PcomA. Another one had an ipsilateral unruptured AchoA aneurysm. Our case showed SAH due to ruptured aneurysm arising from fetal type of PcomA among multiple cerebral aneurysms founded and AchoA originating from ICA proximal to PcomA. The contralateral PcomA and AchoA were normal. Typically, AchoA runs posteromedially within the crural cistern and continues ambient cistern to enter the choroidal fissure. The proximal portion of this artery is cisternal segment and following portion becomes intraventricular segment1). The transposed AchoA in the previous cases including ours did not seem to affect the vascular territory or course despite of unusual vascular arrangements. The AchoA can be exposed or manipulated in surgical approaches into the pathological lesions around temporal lobe or involving supraclinoid ICA. Although the AchoA aneurysms accounting for 4% of all cerebral aneurysms are not so common, vascular injury of AchoA with small caliber supplying the eloquent areas may be critical because of the possibility of significant morbidity and mortality. And, another caution is that AchoA do not have significant collateral flows despite a potential anastomosis with posterior choroidal artery3). We tried to preserve PcomA itself and all perforating branches arising from it because of the possibility to compromise the collateral flows of fetal type PcomA.