A Tale of the Tail : A Comprehensive Understanding of the “Human Tail”
Article information
Abstract
Humans do not have tails; however, a congenital anomaly named “human tail” has been recognized since old times. In contrast with its impactful name, the anomaly itself is not fatal, and thus it has not been considered as a clinically serious symptom. However, many case reports suggested that retention of “the tail” is closely associated with spinal cord malformation and should be treated with care by neurosurgeons. Therefore, this review summarizes our knowledge regarding the anatomy, function, and development of the tail as a general structure in mammals. Learning the basic knowledge regarding tail anatomy and development would help clinicians to understand the “human tail” more concisely and to select more appropriate examinations or treatments in relation to this congenital anomaly.
INTRODUCTION
We, humans, basically do not have tails. However, some clinicians would claim that there is a congenital anomaly named “human tail”. This impressive anomaly has been occasionally reported in clinical case reports, but the anomaly seemed not to be regarded as a serious symptom. This is because the human tail itself is not fatal. Thus, the solid definition of this anomaly has not been established, and the cause has not been clarified yet. But many case reports regarding the human tail showed that this anomaly is highly associated with spinal dysraphism (over 40% of reported cases since 1880s [32]); neurosurgical treatments were required in such cases. Spinal dysraphism is not life threatening, but severe forms such as myelomeningocele, meningocele, lipoma, or tethered cord syndrome, are troublesome anomalies which could affect the patients’ quality of life. This anomaly has been reported all over the world including Asian countries.
This review aimed to provide basic knowledge about the tail anatomy and its development for clinicians to understand this congenital anomaly “human tail” substantially. Although humans basically do not have tails, clinicians may still obtain some benefits to better understand the purpose and structure of tails in other mammals. In fact, learning the basic knowledge regarding tail anatomy and development would help clinicians to understand the congenital anomaly, “human tail,” more concisely and to select more appropriate examinations or treatments.
WHAT IS A TAIL?
In vertebrates, a tail is generally defined as an elongated trunk that is posterior to the anus (or the cloaca). A tail is structurally homologous to the trunk but does not contain a body cavity; it includes musculoskeletal elements (caudal vertebrae and muscles) and neurovascular tissues that are associated with the muscles that move the tail. Tail length varies greatly among mammal species and even among different conspecific local populations [12,35]. It is one of the most important morphological features that reflect the habitat, ecology, and evolutionary processes associated with a given species.
Anatomy of tails
The musculoskeletal system of the tail is presented in Fig. 1. The caudal vertebrae are part of the skeletal system that maintains the tail, which articulates posterior to the sacrum [28]. The morphology of the caudal vertebrae differs between the proximal and distal regions [2,3,13,22,29]. The proximal caudal vertebra, the morphology of which is similar to that of other vertebrae, provides attachments to the caudal musculature or maintains their muscular bellies [22,29]. These vertebrae articulate with one another via their zygapophyses and intervertebral discs, as do other vertebrae. In contrast with the proximal caudal vertebrae, the distal caudal vertebrae are simply shaped in a vaguely hexagonal column. They possess neither processes nor arches and articulate only with the intervertebral discs. These morphological differences between the proximal and distal caudal vertebrae might affect tail mobility. The zygapophyses involved in articulation in the proximal region of the tail permit a certain degree of sagittal flexion but resist movement in other directions [26,27,33]. Conversely, articulation only via the intervertebral discs in the distal region allows movement in almost all directions [22]. Among the taxonomic groups of species that contain tails with varying lengths, the number of caudal vertebrae tends to vary with tail length [11,22,29].

Anatomy of the tail. A : Caudal vertebrae. B : Basic anatomy of caudal musculatures in primates. 1 : M. abductor caudae medialis, 2 : M. abductor caudae lateralis, 3 : M. extensor caudae lateralis, 4 : M. extensor caudae medialis, 5 : M. iliocaudalis+M. pubocaudalis, 6 : M. ischiocaudalis, 7 : M. flexor caudae longus, 8 : M. flexor caudae brevis.
The caudal musculature contains dorsal caudal extensors and abductors and ventral caudal flexors and pelvocaudal muscles. The dorsal caudal muscles are especially considered to be elongated portions of the erector spinae muscles. These structures contribute to the movement of the tail [22,30]. The caudal musculature in tailed primates exists on both the ventral and dorsal sides, as in other mammals, and allows the tail to be flexed, extended, and abducted. The caudal muscles tend to insert more proximally in shorter-tailed primate species. The number of caudal muscles, as well as the number of vertebrae, tends to be reduced in shorter-tailed species [30].
Function of tails
In primates, tails play many important roles. Some New World monkeys (e.g., spider and howler monkeys) have prehensile tails and use them as a “fifth hand”; they can even suspend themselves by their tails. Most primates, however, do not have prehensile tails. Like in most other arboreal mammals, the tail in primates is generally used as a balancer, as supported by experimental studies, to increase stability in the unstable arboreal environment [17]. Indeed, when an individual walks or leaps from branch to branch, the tail whips left and right or rotates. Among the Old World monkeys, arboreal species tend to have longer tails than other species, implying that the need for balance is greater in arboreal species.
Some tails also function in social communication [1,4,14,16,21,23]. For example, tail carriage indicates the rank of a male within the troop hierarchy in some cercopithecoid species, such as the savanna monkey (Cercopithecus aethiops). The alpha male has the highest rank and carries his tail in a sigmoid manner, while the beta male has the second-highest rank and carries his tail parallel to the ground. Similar signaling has been reported in the Japanese macaque (M. fuscata). Tails may also be used for communication between male and female primates. While observing a troop of wild crab-eating macaques (M. fascicularis) in Thailand, I noted some females holding their tails high and showing their vaginas to the alpha male. Immediately after the alpha male saw a female displaying this posture, he mated with her.
EVOLUTION OF TAIL LOSS IN HUMANS
It is striking that an organ with such multiple functions has become completely lost in hominoids (so-called apes), including humans. The tail-reduction process during evolution remains a mystery owing to incomplete fossil records. A common ancestral species of tailed cercopithecoids (Old World monkeys) and tail-less hominoids is Aegyptopithecus zeuxis. This species possessed a distinct distal portion of the caudal vertebra. Thus, we had an ancestor approximately 33 million years ago (Ma) with a long tail [10]. However, more recent ancestral hominoid species (Proconsul heseloni, approximately 18 Ma, and Nacholapithecus kerioi, approximately 15.5 Ma) had already lost their tails completely [19,20,34]. Our ancestors lost their tails during an almost 20-million-year period, between the Oligocene and Miocene epochs. Unfortunately, no fossils connecting these ancestral species have ever been found (Fig. 2).

Tail reduction process during human evolution. A common ancestor species of cercopithecoids and hominoids (Aegyptopithecus zeuxis) possessed a long tail. However, the common ancestral species of hominoids (Proconsul heseloni and Nacholapithecus kerioi) had lost their tails. Humans must also have lost their tails during this period, but no conclusive fossils have been found.
TAIL MORPHOGENESIS DURING EMBRYONIC DEVELOPMENT
Axial extension and termination
The development of the tail is presented in Fig. 3. During embryonic development in amniotes (tetrapod vertebrate species that possess the amnion during development : reptiles and mammals), the body axis elongates and pairs of somites are generated along the axis. These somites are the primordia of the vertebrae and the erector spinae muscles. Somitogenesis in the caudal region proceeds in the same manner with a more anterior trunk : somites are generated sequentially from the presomitic mesoderm along the neural tube [18,24]. When the somites are added, Fgf/Wnt proteins are secreted at the tip of the tail (tail bud). The Fgf/Wnt proteins are antagonistic to retinoic acid (RA) and are present in greater quantity than RA during axial elongation. When the axial elongation terminates, however, these two opposing conditions reverse and the amount of RA at the tail bud increases. New somite formation then stops [6,25].
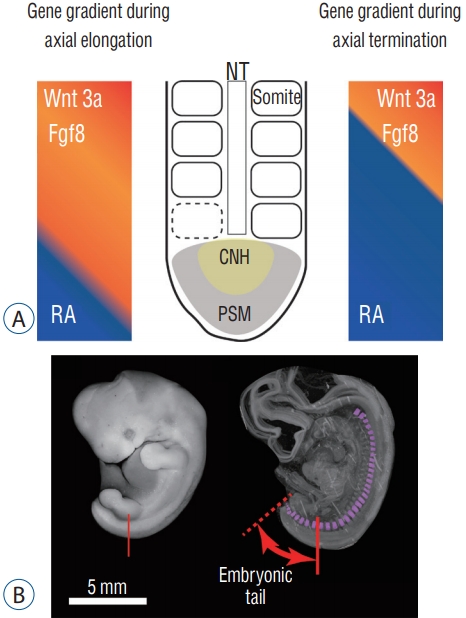
Development of the tail. A : Gradient of gene expression during axial extension and termination. B : Somites in a human embryo. Somites are present up to the tip of the tail. NT : neural tube, RA : retinoic acid, CNH : chordoneural hinge, PSM : presomitic mesoderm.
While the body axis elongates, Hox genes are at work, and variations in their expression characterize the body axis; they determine which embryonic portions become cervical, thoracic, lumbar, and sacrocaudal parts of the body. Studies utilizing mouse models revealed that posterior Hox genes (paralog group 10 to 13) are involved in the determination of tail length in mammals [5]. Another study demonstrated that the number of lumbar and sacral vertebrae was altered in double-knockout Hoxa10 and Hoxd11 mice [9]. Hoxb13 is also related to tail elongation in mice [36]. Another study revealed that a Hoxb13 knockout mouse had a longer tail and a greater number of caudal vertebrae than normal individuals [7].
Tail reduction
Previous studies regarding tail development have suggested that the timing of axial elongation termination is an important developmental event for tail length determination. However, a very recent study showed that another developmental event, tail reduction, is essential in determining the final tail length [31]. It is known that the once-formed embryonic tail diminishes and is completely lost during the human embryonic period. However, until recently, the detailed developmental process behind this event has not been clarified. The authors of a recent study used human embryonic specimens to study the transition of the number of somites and clarified that the caudal somites radically decrease in number immediately after the termination of axial elongation [31]. The cellular behavior and genes involved in this tail reduction have not yet been clarified, but the transition may involve apoptosis [8].
IS “HUMAN TAIL” A REAL TAIL?
The “human tail” is not a fatal anomaly and is usually surgically removed by ablation; however, there have been many instances where the ablations were not treated with care, resulting in complications. Some studies have reported that this anomaly tends to be associated with spinal anomalies, but previous case reports were summarized only partially and locally, and pathological patterns including associated anomalies were not fully understood. Moreover, the definition of the “human tail” itself was inconsistent and its classification varied [32]. The causes of this anomaly have not been studied carefully; it was believed to be a residual embryonic tail for more than 100 years. More specifically, it was believed that there is a region so-called caudal filament which does not contain any somites at the tip of the human embryonic tail [15]. Harrison [15] claimed that the human tail is caused from mal-reduction of the caudal filament.
However, a very recent study using 3D reconstruction of human embryonic structure has shown that somites exists until the tip of the tail and the caudal filament is not observed in the human embryonic tail [31]. This finding denied the 100-year-old Harrison’s hypothesis regarding the cause of the human tail.
Another study analyzed the case reports regarding the human tail and revealed that the cause of the anomaly is multiple though their appearances are similar. The study analyzing 195 cases of “human tail” from 1900 to the present revealed that it can be classified into four types, based on its structure and location [32].
If the tail contains bones or cartilages inside and they are a part of the coccyx, the tail is categorized as typeIa. Though the tail contains a part of vertebrae, they are not excess vertebra and the number of the vertebrae is in the normal range. This type is caused by the mal-curvature or oversize of the coccyx and is not the residual of the embryonic tail. TypeIb tail contains some bony/cartilaginous elements not relevant to the coccyx. This case is highly associated with lipoma or teratoma, and thus these elements seemed to be ectopically formed by those tumors.
When the tail without any bones is located higher than the natal cleft, it is categorized as typeIIa. Among the four human tail types (Ia, Ib, IIa, IIb), this is the most common and neurosurgeons should be most careful to treat this type. It is the most common (92 out of 195 reported cases) and is highly associated with spinal cord malformation (44/92 cases); it is assumed that this could be caused by neural tube closure defects.
The last typeIIb does not contain bones as well and is located within the para-anal region (lower than the natal cleft). Though it is rare, this type of tail occasionally contains striated muscle fibers inside. But these muscle fibers do not originate from the somite (i.e., not related to the erector spinae muscles) but connect to the anus. TypeIIb is often associated with the rectal/anal anomalies, such as imperforate anus, and is possibly the residual of the anal tubercle.
As described, detailed investigation of the case reports, which is based on the basic knowledge of tail anatomy and development, showed us that the cause and appropriate treatment should be different depending on the type, though the symptoms are similar. In addition, this investigation showed that the established belief about the “human tail” for a long period of time is not necessarily correct.
CONCLUSION
Although humans do not have tails, clinicians may still obtain some benefit from understanding the purpose and structure of tails in other mammals. Studying the typical structure, development, and functions of tails improves our knowledge of the world and provides us with perspective on this anatomical feature.
Notes
No potential conflict of interest relevant to this article was reported.
INFORMED CONSENT
This type of study does not require informed consent.
AUTHOR CONTRIBUTIONS
Conceptualization : ST
Funding acquisition : ST
Project administration : ST
Visualization : ST
Writing - original draft : ST
Writing - review & editing : ST
Acknowledgements
I would like to thank Professor Ji Yeoun Lee (Department of Anatomy, Seoul National University College of Medicine) and Professor Chae-Yong Kim (Department of Neurosurgery, Seoul National University College of Medicine) for their invitation to write this review.
This work was supported by JSPS KAKENHI Grant Number JP18K15008.
