Safety and Efficacy of Flow Diverter Therapy for Unruptured Intracranial Aneurysm Compared to Traditional Endovascular Strategy : A Multi-Center, Randomized, Open-Label Trial
Article information
Abstract
Objective
Endovascular treatment of large, wide-necked intracranial aneurysms by coil embolization is often complicated by low rates of complete occlusion and high rates of recurrence. A flow diverter device has been shown to be safe and effective for the treatment of not only large and giant unruptured aneurysms, but small and medium aneurysms. However, in Korea, its use has only recently been approved for aneurysms <10 mm. This study aims to compare the safety and efficacy of flow diversion and coil embolization for the treatment of unruptured aneurysms ≥7 mm.
Methods
The participants will include patients aged between 19 and 75 years to be treated for unruptured cerebral aneurysms ≥7 mm for the first time or for recurrent aneurysms after initial endovascular coil embolization. Participants assigned to a flow diversion cohort will be treated using any of the following devices : Pipeline Flex Embolization Device with Shield Technology (Medtronic, Minneapolis, MN, USA), Surpass Evolve (Stryker Neurovascular, Fremont, CA, USA), and FRED or FRED Jr. (MicroVention, Tustin, CA, USA). Participants assigned to a coil embolization cohort will undergo traditional endovascular coiling. The primary endpoint will be complete occlusion confirmed by cerebral angiography at 12 months after treatment. Secondary safety outcomes will evaluate periprocedural and post-procedural complications for up to 12 months.
Results
The trial will begin enrollment in 2022, and clinical data will be available after enrollment and follow-up.
Conclusion
This article describes the aim and design of a multi-center, randomized, open-label trial to compare the safety and efficacy of flow diversion versus traditional endovascular treatment for unruptured cerebral aneurysms ≥7 mm.
INTRODUCTION
The prevalence of intracranial aneurysms is 0.4–3.0% [25,28], and aneurysmal rupture results in subarachnoid hemorrhage that has a decimal prognosis with substantial mortality and morbidity [4,28,29]. Advances in non-invasive brain imaging methods such as magnetic resonance angiography have facilitated the diagnosis of unruptured intracranial aneurysms [24], and the preventive treatment of unruptured intracranial aneurysm is generally recommended for an aneurysm with a diameter ≥7 mm [25].
Microsurgical clipping and endovascular coil embolization are two major treatment modalities for intracranial aneurysms [5]. Equipped with the advantage of non-invasiveness and advances in devices and techniques, coil embolization has become the dominant treatment choice with lower procedural morbidity and mortality [3,12,18]. However, coil embolization of wide-necked aneurysms is still difficult; even with adjunct use of stent, recurrence and retreatment rates are approximately 12% and 6%, respectively [7,15]. Furthermore, larger aneurysms are associated with higher rates of recurrence even when complete or near-complete occlusion has been initially achieved, due to coil compaction leading to recanalization [10,11,17]. A study revealed that large aneurysms (≥7 mm) were more likely to recur than smaller aneurysms (<7 mm) after coil embolization [26].
Flow diversion has been developed to address the challenges faced during traditional coil embolization. Flow diverters are designed to exert lower radial force to improve conformability and provide increased metal surface coverage for improved flow diversion, compared to conventional intracranial stents [27]. They elicit remodeling of the parent artery primarily through the following mechanisms : 1) exclusion of blood flow into the aneurysmal sac, resulting in thrombus formation inside the sac and 2) provision of a scaffold for endothelialization [23]. A clinical trial using the Pipeline embolization device (Medtronic, Minneapolis, MN, USA), the first commercially available flow diverter, revealed that flow diversion of large aneurysms (>10 mm) in the internal carotid artery with wide neck (>4 mm) showed a complete occlusion rate of 86% at 12 months, and major morbidity and mortality rate of 5.6% [1], and the device was approved by the United States Food and Drug Administration (FDA) for use in large aneurysms (>10 mm) with wide neck (>4 mm). Subsequently, a recent clinical trial using the Pipeline Flex embolization device for unruptured wide-necked aneurysms ≤12 mm demonstrated complete occlusion rate of 76.8% at 12 months with a major morbidity and mortality rate of 2.1% [13]. The U.S. FDA has expanded the instructions for its use in wide-necked aneurysms with a small- and medium-sized aneurysms.
In Korea, the flow diverter was initially approved for use in the treatment of unruptured aneurysms ≥15 mm, and only recently was it approved for aneurysms ≥10 mm. Currently, endovascular treatment options for wide-necked aneurysms <10 mm are limited to traditional procedures such as parent artery occlusion, coil embolization by multiple-catheter technique, or balloon or stent-assist technique.
The aim of this study is to assess the safety and efficacy of flow diversion treatment of unruptured intracranial aneurysms ≥7 mm by evaluating complication rates and complete occlusion rates at 12 months compared to traditional endovascular treatment in patients who are either to receive the first treatment or retreatments for recurrent aneurysms after initial endovascular treatment.
MATERIALS AND METHODS
Design
This is a prospective, multicenter (13 institutions), randomized, open-label trial to compare the safety and efficacy of treatment of unruptured cerebral aneurysms or recurrent cerebral aneurysms with a maximum diameter ≥7 mm by flow diversion versus coil embolization with or without adjunctive balloon or stent devices. Approval of the institutional review board at each participating institution has been obtained (see Supplementary Data 1). Participants will be randomly assigned to flow diversion or traditional endovascular treatment cohorts in a 1 : 1 fashion. The primary endpoint will be complete occlusion confirmed by catheter cerebral angiography at 12 months after endovascular treatment.
Study population
The study population will include patients aged between 19 and 75 years diagnosed with unruptured intracranial aneurysms or with recurrent aneurysms after initial endovascular treatments with a maximum diameter ≥7 mm arising from the internal carotid artery, anterior cerebral artery, middle cerebral artery, or vertebrobasilar arteries. Recurrent aneurysms are defined as grade 2 (70% to 89% aneurysm occlusion in orthogonal images of catheter cerebral angiography from optimal projection) or higher according to the aneurysm occlusion grading system for endovascular aneurysm occlusion suggested by Meyers et al. [16] (Table 1). Detailed inclusion and exclusion criteria of the study are described in Table 2.
Informed consent
The informed consent form will contain extensive information about this trial including the purpose of the trial, the study design, potential risks and benefits associated with the trial, and the participants’ rights and responsibilities.
Randomization
After complete explanation of the trial and signing informed consent, participants will be enrolled to the trial and randomized to either the flow diversion or coil embolization cohort in a 1 : 1 ratio using a computer-generated randomized sequence. For allocation concealment, central randomization will be performed, and a random number table will be kept by a third party other than the investigators. The participant will be informed of the treatment he/she is assigned when signing the informed consent for the treatment.
Treatment
Patients assigned to flow diversion will undergo placement of any of the following flow diverters available in Korea : Pipeline Flex Embolization Device with Shield Technology (Medtronic), Surpass Evolve (Stryker Neurovascular, Fremont, CA, USA), and FRED or FRED Jr. (MicroVention, Tustin, CA, USA). Participants assigned to coil embolization will undergo endovascular coiling of aneurysms with or without adjunctive balloon or stent devices. Dual antiplatelet agents comprising of aspirin 100 mg and clopidogrel 75 mg will be administered to all participants 5 to 7 days prior to the procedure and continued for a maximum of 12 months, after which aspirin monotherapy will be maintained for at least 2 years after the procedure.
Clinical and radiologic outcome measures
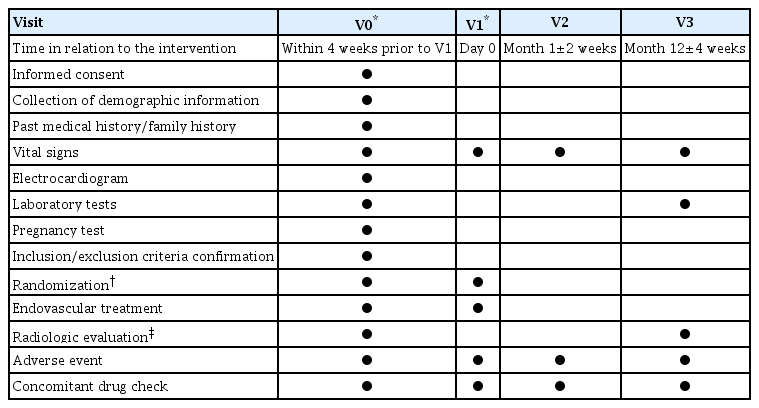
The primary endpoint will be complete occlusion (grade 0 or 1 confirmed by catheter cerebral angiography according to the Aneurysm occlusion grading system) at 12 months after the treatment. Secondary endpoints will evaluate the short-term and the longer-term safety of the trial, and include the following : 1) any procedure-related complications defined as thromboembolic or hemorrhagic complications that occur within 30 days after the treatment, 2) severity of hemorrhage evaluated by Thrombolysis in Myocardial Infarction bleeding criteria [8], 3) delayed intracranial hemorrhage defined as spontaneous intracranial hemorrhage that occurs within 12 months after treatment, 4) delayed cerebral infarction defined as cerebral infarction due to thromboembolism within 12 months after treatment, and 5) neurologic status evaluated using the modified Rankin scale score at 1 month and 12 months. A summary of the scheduled visits is shown in Table 3.
Sample size
The primary endpoint will be the complete occlusion of the aneurysm at 12 months after the endovascular treatment. Based on data from previous studies, we estimate the longterm complete occlusion rate of cerebral aneurysms ≥7 mm to be 80% [9,14,19]. The complete occlusion rate of recurrent aneurysms treated with flow diversion was 94.1% [20], and considering the improvements in device and technique, we estimate the complete occlusion rate of cerebral aneurysms using flow diverters to be 95%, yielding a risk difference of 15%. Taking into account a significance level of 0.05, and power of 80% for the two-sided chi-square test, the number of participants required is 76 per group. Based on our previous experience in which drop-outs of cerebral aneurysm patients were relatively rare, we expect a patient dropout rate of 5%, yielding the total number of patients required for the study to be 160 (80 patients per group).
Statistical analysis
The principal method of analysis is intention-to-treat analysis and a full analysis set will be used for statistical analysis, but per-protocol analysis may also be performed, as flow diverters may not be feasible in cases with unfavorable complex vascular anatomy. For bivariate analysis, a two-sample t-test or Mann-Whitney U test will be performed for continuous variables, and the chi-square test or Fisher’s exact test will be used for categorical variables. The primary and secondary outcomes will be evaluated by logistic regression analysis, and the effect size will be presented as odds ratios and 95% confidence intervals.
RESULTS
The trial will begin enrollment in 2022, and clinical data will be available after enrollment and follow-up.
DISCUSSION
This study aims to provide definitive data on the safety and efficacy of flow diversion with devices available in Korea in comparison to traditional endovascular treatment for unruptured cerebral aneurysms ≥7 mm.
Previously, several studies have compared the treatment outcomes of flow diversion and coil embolization, but they were retrospective studies. A systematic review and metaanalysis by Zhou et al. [32] reported that flow diversion showed a significantly higher complete occlusion rate than coil embolization and a subgroup of stent-assisted coil embolization. A recent meta-analysis comparing flow diversion treatment with conventional endovascular treatment revealed that flow diversion was associated with higher 6-month complete occlusion rate for large or giant aneurysms, but no such difference was observed for non-large or giant aneurysms [30]. The current randomized trial is expected to minimize selection bias and confounding factors due to unequal distribution of significant factors, and to warrant direct comparison of the two treatment methods.
The off-label use of flow diverters for aneurysms <10 mm has already been prevalent, and its efficacy and safety have also been reported for different flow diverters such as FRED (MicroVention) [2,21] and SILK (Balt Extrusion, Montmorency, France) [22]. A meta-analysis of flow diversion treatment for intracranial aneurysms <10 mm revealed the complete occlusion rate of 84.2%, and procedure-related mortality and morbidity of 0.9% and 5.2%, respectively [31]. A matched-cohort study by Chalouhi et al. [6] demonstrated that flow diversion was associated with a significantly higher complete occlusion rate than coil embolization (70% and 47.5%, respectively, p=0.04) without additional morbidity in the treatment of small aneurysms <10 mm. To date, no study has directly compared the treatment outcomes of flow diversion and traditional endovascular coiling for treatment of aneurysms <10 mm.
CONCLUSION
This article describes the design of a multi-center, randomized, open-label trial to compare the safety and efficacy of the treatment of unruptured cerebral aneurysms with maximum diameter ≥7 mm by flow diversion versus traditional endovascular treatment. This trial is expected to warrant direct comparison of the two treatment methods with minimal bias.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Informed consent
This type of study does not require informed consent.
Author contributions
Conceptualization : JK, GH, BTK, SQP, JSO, SPB, OKK, JC, Committee of Multicenter Research, Korean Neuroendovascular Society; Funding acquisition : OKK, JC; Methodology : JK, GH, BTK, SQP, JSO, SPB, OKK, JC, Committee of Multicenter Research, Korean Neuroendovascular Society; Project administration : JK, GH, BTK, SQP, JSO, SPB, OKK, JC, Committee of Multicenter Research, Korean Neuroendovascular Society; Writing - original draft : JK; Writing - review & editing : OKK, JC
Data sharing
None
Preprint
None
Acknowledgements
This research is supported by Korean NeuroEndovascular Society research fund (Kones-2020-02) and a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the ministry & Welfare, Republic of Korea (grant number : HC20C0057). We also would like to thank Editage (www.editage.co.kr) for English language editing.
Trial Registration : Clinical Research Information Service Identifier (KCT0006474).
Supplementary materials
The online-only data supplement is available with this article at https://doi.org/10.3340/jkns.2022.0043.