What Should Be Considered to Cause the Early Post-Craniotomy Seizure: Antibiotics (Cefazolin) Irrigation
Article information
Abstract
Objective
Post-craniotomy seizure (PCS) is reported only rarely. However, our department noted a 433% increase in PCS for a year beginning September 2010, especially after cerebrovascular surgery. Our goal was to identify the cause of our unusual outbreak of PCS.
Methods
For almost one year after September 2010, cases of PCS increased significantly in our department. We analyzed 973 patients who had received a major craniotomy between January 2009 and November 2011. We included seizures that occurred only in the first 24 postoperative hours, which we defined as early PCS. After verifying the presence of PCS, we analyzed multiple seizure-provoking factors and their relation to the duration and character of seizure activity.
Results
Overall PCS incidence was 7.2% (70/973). Cefazolin (2 g/L saline) was the antibiotic drug used for intraoperative irrigation in 88.4% of the operations, and no PCS occurred without intraoperative cefazolin irrigation. When analyzed by operation type, clipping surgery for unruptured aneurysms was the most frequently associated with PCS (80%). Using logistic regression, only 2 g cefazolin intraoperative irrigation (p=0.024) and unruptured aneurysm clipping surgery (p<0.001) were associated with early PCS. The seizure rate of unruptured aneurysm clipping surgery using 2 g cefazolin intraoperative irrigation was 32.9%.
Conclusion
Intraoperative cefazolin irrigation must be avoided in patients undergoing craniotomy, especially for clipping of unruptured aneurysms, because of the increased risk of early PCS.
INTRODUCTION
In patients who have a neurosurgical infection, intraventricular or intravenous treatment with antibiotics such as penicillin, gentamicin, cephalothin, cefazolin, and vancomycin is common practice681419). However, in the 1980's, some studies documented that a few antibiotics were associated with postoperative seizure. For example, Wald and McLaurin19) showed that intraventricular cephalothin caused seizures, but not gentamicin or methicillin. And Park and Lee17) demonstrated that cortical application of some antibiotics could lead to a variety of complications of immediate or delayed seizure, cortical electrical depression, radiculopathy, and transverse myelopathy. In addition, Michenfelder et al.15) insisted that intraoperative, intravenous (i.v.) penicillin administration should be avoided in patients undergoing craniotomy because of the potential for i.v. penicillins (but not non-penicillins) to induce early post-craniotomy seizures (PCS).
Despite these data, no definitive evidence specifically regarding cefazolin was published, although there are a few clinical and animal experimental reports. Thus, our neurosurgical department has routinely used i.v. administration of antibiotics (usually cefazolin) as a routine procedure within one hour of the start of surgery. In addition, our use of intraoperative saline irrigation mixed with antibiotics (usually cefazolin, 1 g/L saline) has been a common procedure for 10 years, given that we saw no problems from cefazolin irrigation, only the anti-infection advantages of antibiotics. However, since September 2010, there has been an outbreak of PCS in our department, so we undertook a close investigation of the cause of the seizures by using a retrospective review and analyzing some risk factors related to PCS.
MATERIALS AND METHODS
Our hospital Institutional Review Board approved this retrospective study. Using the September 2010 date as a basis, we included 973 patients who underwent major craniotomy from January 2009 to November 2011. We excluded the revision craniotomies, such as tumor recurrence or postoperative hemorrhage, and also excluded patients with postoperative pentobarbital coma therapy, because both tumor and hemorrhage can influence PCS, and pentobarbital coma therapy can mask seizure activity. We only included seizures occurring in the first 24 postoperative hours as being seizures related to craniotomy (PCS). The following data were abstracted from each medical record : age, sex, underlying disease, history of preoperative seizures, type of operation, site of operation, main operation time, primary anesthetic agent, type of intravenous antibiotics, type of antibiotics for intraoperative irrigation, type of muscle relaxant, type of prophylactic seizure medication, and type of seizure in the first 24 postoperative hours.
We have routinely used prophylactic anti-epileptic drugs (valproic acid) for all supratentorial craniotomies, but not for infratentorial ones. Intraoperative irrigation with the saline solution mixed with cefazolin was always performed at the end of brain surgical procedures roughly with the amount of 1 L. Though our standard practice for intraoperative cefazolin irrigation was to use the stock concentration of 1 g cefazolin/L saline (we performed intra-operative irrigation using cefazolin mixed 1 L fluid after main procedure), the cefazolin antibiotic stock concentration changed from 1 g to 2 g around September 2010. At this time we decided to increase the dosage of antibiotics in a saline solution, expecting the high concentration of antibiotics to be more effectively preventive of infection as generally accepted. Thus, our intraoperative cefazolin irrigation concentration also changed from 1 g/L cefazolin to 2 g/L cefazolin.
Standard statistical analysis was performed using SPSS version 18.0 (SPSS Inc., Chicago, IL, USA). Differences between groups were assessed by using the chi-square (χ2) test and Spearman rank correlation. A logistic regression analysis was also performed. We rejected null hypotheses (i.e., achieved statistical significance) with a p value of less than or equal to 0.05.
RESULTS
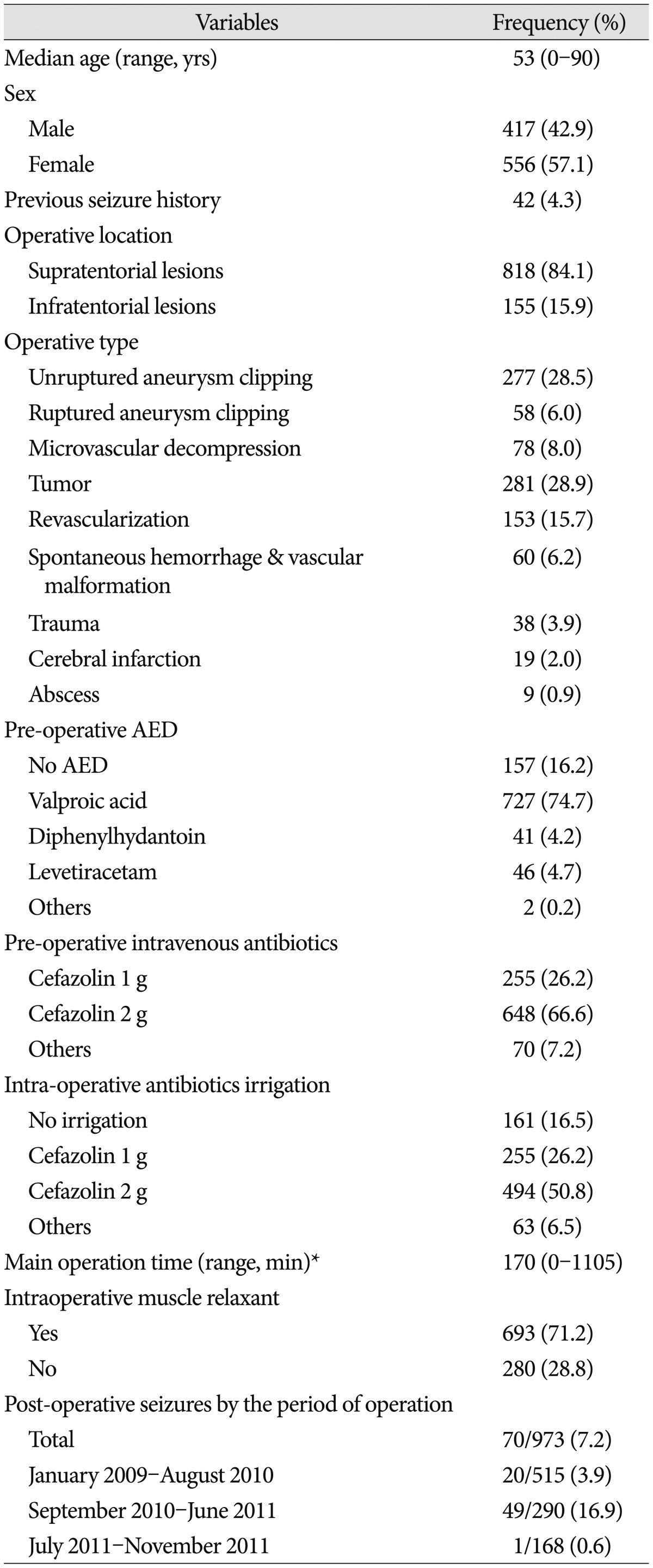
Table 1 shows the general characteristics of all 973 patients. Overall, the PCS rate was 7.2% (70/973). From January 2009-August 2010, when 1 g/L cefazolin was used for intraoperative irrigation, the seizure rate was 3.9%. However, from September 2010 to June 2011, when 2 g/L cefazolin was used, the seizure rate increased to 16.9%. Surprisingly, nearly no seizure activity (0.6%) occurred during the period after July 2011, when cefazolin irrigation was discontinued (Table 1). The seizure proportion based on the intraoperative irrigation of antibiotics was the highest (87.2%, n=61) with 2 g/L cefazolin, though even higher with 1 g/L cefazolin (10.0%, n=7) than without antibotics (Table 2). Clipping surgeries for unruptured aneurysms were the type of surgeries most associated with PCS (80%). In addition, microvascular decompression (MVD), despite being an infratentorial craniotomy, was associated with PCS with the rate of as much as 5.7% (Table 2). A total of 70 seizures occurred within the first 6 postoperative hours, and the median seizure-onset time after dura mater closure was 90 minutes. There were 48 cases (68.6%) of generalized-type seizures, and in all but two patients, seizure activity, as demonstrate by electroencephalogram, terminated within 12 to 24 hours postoperatively. In two patients, sedative treatment with midazolam/propofol was used postoperatively over 48-72 hours while monitoring the electroencephalogram due to status epilepticus. The general characteristics of all seizure patients are shown in Table 2.
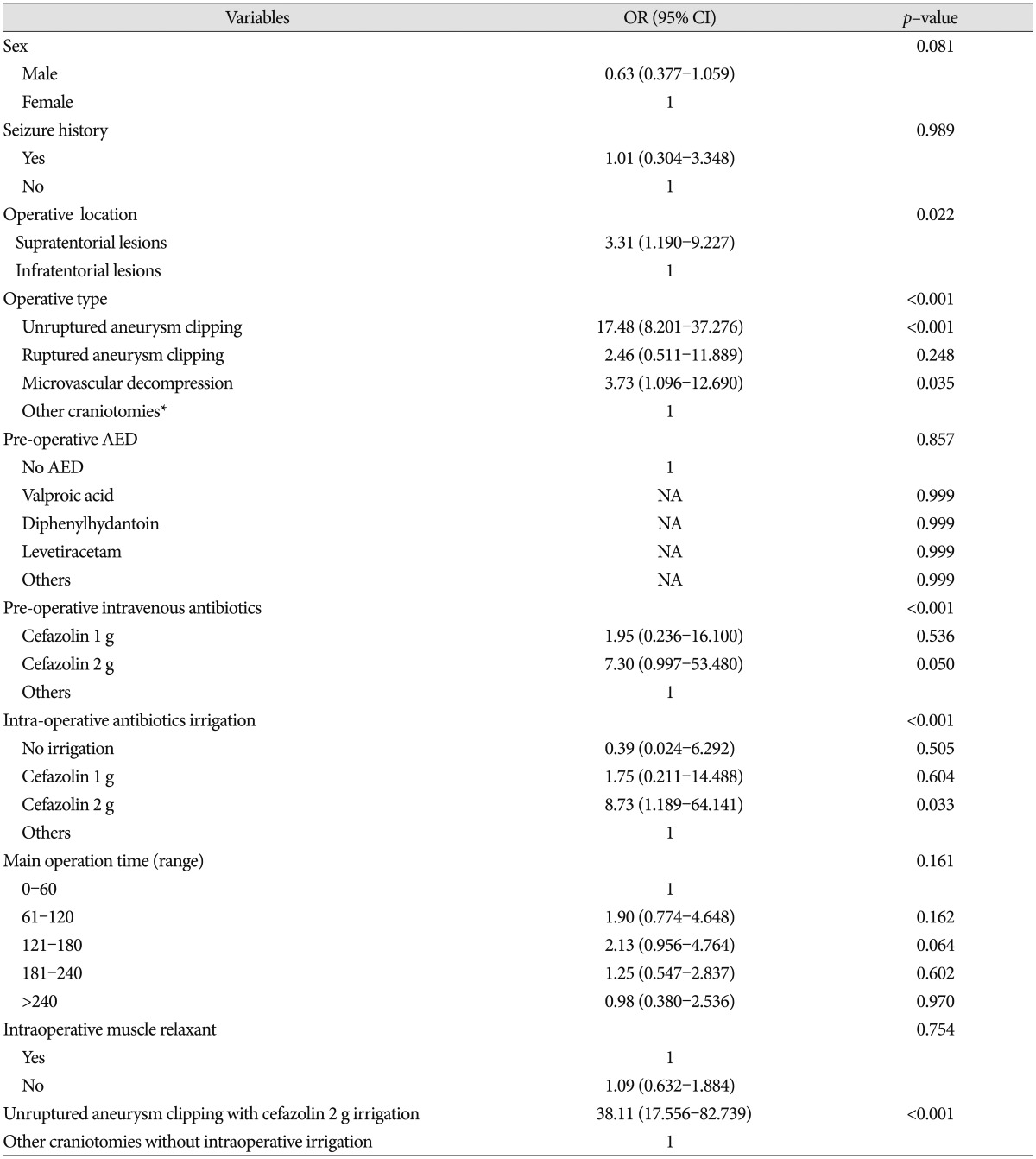
In univariate logistic regression analysis of all factors, hyperlipidemia, location of operation, operation type, and antibiotic use were associated with PCS (Table 3). Intraoperative antibiotic irrigations had been used with preoperative intravenous injection of antibiotics (cefazolin 1 g) until June 2011, but the irrigations (not the i.v. antibiotics) were discontinued from July 2011 to November 2011. In that period, only one seizure (0.6%) occurred, so we were able to conclude that intraoperative cefazolin irrigation (2 g/L greater than 1 g/L) was the direct cause of PCS. We therefore could exclude intravenous cefazolin (1 g) injection as a risk factor for PCS.
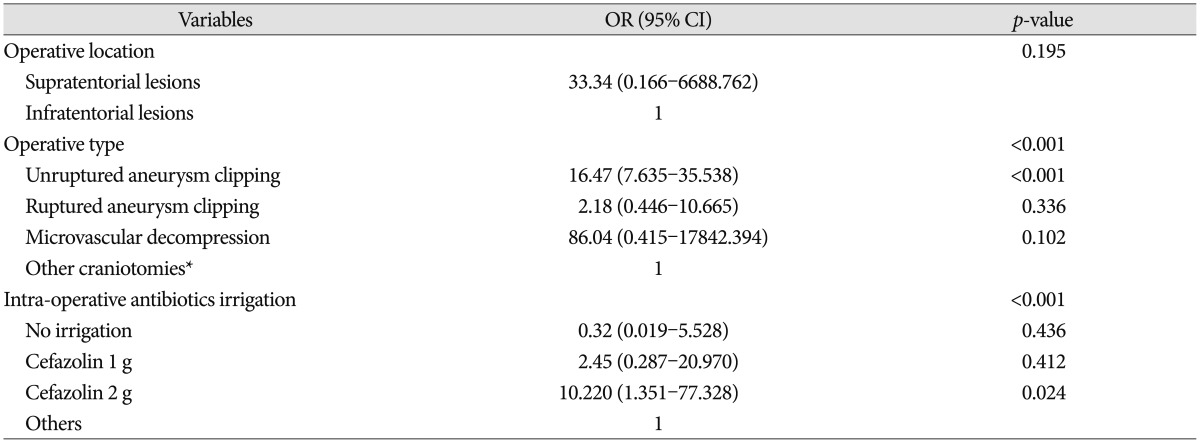
In logistic regression analysis of the factors including hyperlipidemia, location of operation, operation type, and intraoperative antibiotics irrigation, only 2 g cefazolin intraoperative irrigation (p=0.024) and unruptured aneurysm clipping (p<0.001) were independently associated with PCS (Table 4). The seizure rate of the unruptured aneurysm clipping operation type, when 2 g/L cefazolin intraoperative irrigation was used, was 32.9%. Although not statistically significant, the seizure rate of MVD operations with 2 g/L cefazolin irrigation was 5.5%, whereas no seizures occurred in the other infratentorial craniotomies (Table 2). Fortunately, no neurological sequelae resulted from any of the 70 post-craniotomy seizures.
DISCUSSION
This retrospective survey supports the hypothesis that intraoperative cefazolin irrigation causes PCS. Over one year, we experienced a dramatic increase in the PCS rate when the dosage of cefazolin doubled from 1 g/ampule to 2 g/ampule (per 1 L saline bottle) during surgery. At that time, nobody in our department paid attention to the changed cefazolin ampules. Although the epileptogenic effect was not transparent with the use of 1 g cefazolin/L saline, the effect became apparent upon doubling of the cefazolin dosage (2 g/L saline). Surprisingly, PCS rarely occurred in tumor surgeries relative to aneurysm clipping surgery. Also unusual was the 5.5% seizure rate of patients receiving infratentorial MVD. One notable similarity between clipping and MVD is that the surgical work is done in the cisternal space, as opposed to tumor surgery, in which surgical activity is performed mainly within brain parenchyma. This connection suggests that PCS seems to be related to opening of the cisternal space, a part of the irrigation procedure, so we concluded that intraoperative irrigation could be related to PCS.
Reports produced since the 1970's documented that perioperative antibiotics could cause seizures. The most epileptogenic compounds were the beta-lactam antibiotics such as penicillin, cefazolin, and cabapenem145791113162021). Though penicillin's effects were documented in human clinical experiences, for cefazolin most data were from animal models15). Few reports provided clinical cases about cefazolin-induced seizure231012). Schliamser et al.18) tried to identify the pathogenesis of beta-lactam epileptogenesis based on previous reports, identifying excessive doses, decreased renal function, damage to the blood-brain barrier, and concurrent use of nephrotoxic or seizure threshold-lowering drugs as the main factors. Most of the previous articles concluded that perioperative i.v. beta-lactam administration should be avoided. However, our analysis demonstrates that perioperative i.v. cefazolin administration appears safe, except in patients with predisposing factors such as renal disease, given that no seizures occurred after exclusive use of 1 g/L i.v. cefazolin. Careful i.v. use is valuable because cefazolin is one of the best prophylactic antibiotics for surgery. On the other hand, intraoperative cefazolin irrigation, especially with 2 g/L cefazolin, appears neurotoxic and should be avoided in craniotomy.
The main limitation of this study is that it is a retrospective analysis; however, one factor was manipulated deliberately : cefazolin irrigation was purposely suspended from July 2011, and the link between cefazolin irrigation and PCS was thus confirmed.
CONCLUSION
After systematically eliminating other suspicious factors, we conclude that intraoperative cefazolin irrigation must be avoided in patients undergoing craniotomy.
Acknowledgements
This study was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI10C2020).