Risk Factors of Rehemorrhage in Postoperative Patients with Spontaneous Intracerebral Hemorrhage : A Case-Control Study
Article information
Abstract
Objective
Rehemorrhage is the most severe complication of postoperative patients with spontaneous intracerebral hemorrhage. The aim of the present study was to assess independent predictors of rehemorrhage and find the possibility of preventing rehemorrhage in postoperative patients with spontaneous intracerebral hemorrhage (sICH).
Methods
Medical records of 263 postoperative patients with sICH from our Hospital were reviewed. The relationships between rehemorrhage and parameters were examined by univariate and multivariate analyses. The parameters include time from onset to surgery, hematologic paremeters, neuroimaging characteristics, level and variability of systolic blood pressure, medical histories, operation duration, and blood loss. In addition, relationship between rehemorrhage and clinical outcome were analyzed by using multivariate analyses.
Results
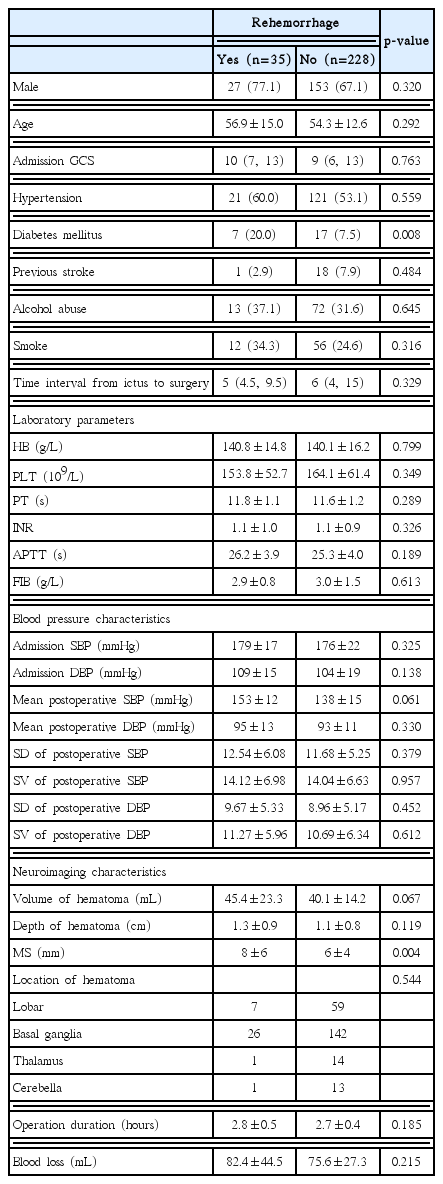
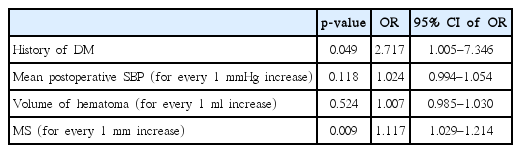
Thirty-five (13.31%) patients experienced rehemorrhage after operation. Multivariate analyses indicated that the following factors were independently associated with rehemorrhage : history of diabetes mellitus (odds ratio [OR], 2.717; 95% confidence interval [CI], 1.005–7.346; p=0.049), and midline shift (for every 1 mm increase, OR, 1.117; 95% CI, 1.029–1.214; p=0.009). Rehemorrhage was an independent risk factor of poor functional outcome (OR, 3.334; 95% CI, 1.094–10.155; p=0.034).
Conclusion
Our finding revealed that history of diabetes mellitus and admission midline shift were possibly associated with rehemorrhage in postoperative patients with sICH.
INTRODUCTION
Spontaneous intracerebral hemorrhage (sICH) accounts for 10–15% of all kinds of stroke4,15). It is reported that the 30-days mortality of sICH is about 30–55% and only 12–39% of the survivors have favorable functional outcomes at 6 month16,24,30).
Treatment of sICH is still debatable among surgeons up to now. One heated topic of the controversy is whether evacuation of hematoma by surgery would be able to improve the prognosis of patients17,18,23). Even so, quite a number of patients had to receive surgery treatment to reduce the intracranial pressure (ICP) because of the relatively huge volume of hematoma. Rehemorrhage is the most severe complication of postoperative patients with sICH. It was reported that the incidence of rehemorrhage is about 11–36% after surgery in patients with sICH and only few patients could have a favorable outcome19,23). Most of previous studies focused on predictors of hematoma growth instead of rehemorrhage. These studies found that level of blood pressure (BP), variability of BP, low levels of fibrinogen, prothrombin complex and alcohol consumption might be associated with hematoma enlargement6,7,12,28). However, using these predictors to evaluate the risk of rehemorrhage in postoperative patients directly might not be appropriate because of hemodynamic change, decreased ICP and brain tissue injury. Some previous studies showed that the time interval between ictus and surgery, surgery methods, and BP might be associated with rehemorrhage in postoperative patients with sICH, but they did not examine the interrelationships among those predictors5,19). In order to assess independent predictors of rehemorrhage and to find the possibility of preventing rehemorrhage in postoperative patients with sICH, here we present this retrospective study.
MATERIALS AND METHODS
This study was a retrospective case control study and got the approval from Biological and Medical Ethics Committee (BMEC) of West China Hospital. The patients’ consent was exempted by the BMEC for the present study was a retrospective clinical study.
Medical records of the patients with sICH between December 2011 and February 2013 from our center were reviewed. Adult patients with sICH who met the indications of surgery and received surgical treatment were eligible. Patients were excluded if : secondary intracerebral hemorrhage (intracranial tumor, arteriovenous malformation or aneurysm); dementia or disability existed before sICH happened; too much intraoperative bleeding caused hemoglobin less than 70 g/L and coagulation disorders or infusion of allogeneic blood whenever it happened; multiple intracerebral hemorrhages, with coagulation disorders or history of anticoagulant drug, with severe hepatic and renal dysfunction.
Indications of surgery were : 1) meet the diagnostic criteria of the American Heart Association (AHA)/American Stroke Association (ASA) intracerebral hemorrhage guidelines (2010)20); and 2) the computed tomography (CT) scan shows the volume of hematoma is above 30 mL in supratentorial region or above 10 mL in cerebellum or indications of intracranial hypertension.
Contraindications of surgery were : 1) with terminal brain hernia (bilateral pupils dilatation and central respiratory-cycle failure).
After hospitalization, all the patients were managed in the stroke unit and followed the recommendations of AHA/ASA guidelines20). Medical history and neurologic physical examination were recorded immediately. CT-scan and routine laboratory examinations including blood routine examinations, biochemical examinations and coagulation studies were performed at the same time. Midline shift applied to supratentorial hemorrhage, which is based on preoperative CT-scan. All the patients had their vital signs monitored. The supportive treatments were given simultaneously. After preoperative preparation finished, all the patients received hematoma evacuation by craniotomy, or minimally invasive surgery (MIS) including neuroendoscopy and stereotactic aspiration. For the uncertainty of efficacy of these approaches, the choice of them mostly depended on surgeon’s preference. The only criterion noted is that if ICP elevated rapidly, such as unilateral pupil dilated, stereotactic aspiration would be the first choice. All the surgeries were conducted by well-trained neurosurgeons (H.L. and C.Y.). The clinical experience of each of them was more than 15 years. Patients were sent to stroke unit or neurological intensive care unit and received standardized treatments after surgery. The postoperative treatments included airway management, BP management (systolic blood pressure [SBP]<180 mmHg), supportive treatments, and other individual treatments.
The volume of hematoma is calculated by the formula A×B×C/2, where A is the greatest diameter on the largest hemorrhage slice, B is the maximal diameter perpendicular to this, and C is the vertical hematoma depth11). Conventional CT-scan was performed at admission, 1 day, 3 days, and 7 days. Besides, unplanned CT scan was performed if necessary. Rehemorrhage was confirmed if postoperative hematoma volume was greater than the preoperative volume or postoperative hematoma volume was smaller than the preoperative volume, but the difference less than 5-mL19,23). In this study, rehemorrhage was also considered if postoperative hematoma volume gradually increased by 10 mL. All the patients were followed-up for at least 3 months. The clinical outcome was evaluated by using the modified Rankin Scales (mRS). The 3-months mRS was dichotomized as favorable outcome (mRS 0 to 2) and poor outcome (mRS 3 to 6).
In the present study, both level and variability of BP were analyzed. Levels of BP were measured on admission, during surgery by the arterial line, every 15 minutes during the first 24 hours after surgery, and every 60 minutes during the next few days. Mean postoperative BP was the mean of successive BP values measured every 3 hours after surgery and before rehemorrhage or 7 days after surgery (or hospital discharge if this occurs earlier). The variability of BP was evaluated by standard deviation (SD) (
All the data were analyzed by using SPSS version 21.0 (IBM Corp., Armonk, NY, USA) and EXCEL 2010 (Microsoft, Redmond, WA, USA). Statistically significance was assumed with a probability value of less than 0.05. The t-test was used to assess the differences in hematologic parameters, BP level, neuroimaging characteristics, operation duration and blood loss between the patient groups with and without hematoma growth. Continuous values were expressed as mean and SD. Discontinuous variable data, such as time interval, were expressed as median and interquartile range and analyzed by Wilcoxon rank sum test. A chi-square analysis was used to test the association between rehemorrhage and location of hematoma. Chi-square test was also used for analyzing the other categorical values. The factors with a probability of <0.1 were entered into a multivariate logistic regression analysis to determine adjusted ORs. The multivariate logistic regression analysis was also used for adjusting the baseline imbalance in the comparison of functional outcome between the patients with and without rehemorrhage.
RESULTS
From December 2011 and February 2013, 561 patients were screened and 281 patients meet the inclusion criteria of this study. Of the 281 patients, 18 patients lost to follow up until 3 months and were excluded. All of them were not rehemorrhage within 7 days after surgery. The mean ages of the patients were 54.6±12.9 years old. Of all the hematomas, 168 (63.88%) located at basal ganglia, while 66 (25.10%), 15 (5.70%), and 14 (5.32%) located at lobar, thalamus, cerebellum respectively. Of all the patients, 192 (73.00%) patients received hematoma evacuation by craniotomy, and the rest 71 (27.00%) patients received hematoma evacuation by MIS. 35 rehemorrhage cases were found 24 (24/192, 12.5%) in craniotomy group and 11 (11/71, 15.5%) in MIS group, and there was no statistic difference (p=0.526). The median time interval between sICH onset and surgery is 6 hours (4, 14).
In the present study, 35 patients (13.31%) experienced rehemorrhage in 7 days after surgery. The mean volume of rehemorrhage hematoma was 26.7±7.7 mL, and the mean time of rehemorrhage after surgery was 4.1±1.7 days. The characteristics of the group with rehemorrhage and group without rehemorrhage were compared in Table 1. Of all the parameters, only the history of diabetes mellitus and midline shift seem to be related to rehemorrhage significantly. Volume of hematoma and mean postoperative BP tended to be higher in the groups with rehemorrhage. There were no differences in other variables including time interval from ictus to surgery and laboratory examinations. At 3 months, 75 (28.52%) had a favorable functional outcome and the rest 188 (71.48%) patients had a poor functional outcome at 3-months post ictus. Of the 35 patients with rehemorrhage, only 4 had a favorable functional outcome. After adjust the age, admission Glasgow coma scale and volume of hematoma, the multivariate logistic regression revealed that the rehemorrhage was an independent predictor of poor functional outcome (OR, 3.334; 95% CI, 1.094–10.155; p=0.034).
Mean postoperative SBP, history of diabetes mellitus, midline shift, and volume of hematoma were selected as possible risk factor for rehemorrhage. Multivariate logistic regressions showed that the history of diabetes mellitus and midline shift were associated with rehemorrhage independently (Table 2).
DISCUSSION
In the present study, the associations between rehemorrhage and some important risk factors were examined in postoperative patients with sICH. The multivariate analyses revealed that history of diabetes mellitus and midline shift were independent predictors of rehemorrhage. In addition, after adjusted some risk factors, we found that the rehemorrhage was independent predictors of poor functional outcome.
Previous studies found that the coagulation dysfunction is associated with hematoma growth after ictus. Patients with low levels of platelets and fibrinogen appear to have a higher risk of hematoma enlargement7). Hypothetically, rehemorrhage after surgery might be related to coagulation disorders as well. In this study, we excluded patients with coagulation disorders before surgery because of the risk of intraoperative bleeding. This might be the reason why there was no significant difference in admission laboratory parameters between the two groups. It was confirmed that loss of blood during surgery might affect the coagulation function33). So we also excluded the patients who underwent too much intraoperative bleeding that caused hemoglobin less than 70 g/L and coagulation disorders or infusion of allogeneic blood whenever it happened.
The patients who suffered multiple intracerebral hemorrhages or with coagulation disorders or history of anticoagulant drug or with severe hepatic and renal dysfunction would place an ICP monitor and give conservative treatment, including mannitol, head elevation, dialysis, infuse the plasma to correct coagulation disorders and so on, to reduce the ICP and stabilize the condition. If the ICP remained above 25 mmHg for 1 to 12 hours or unilateral dilated pupils appeared, decompressive craniectomy would be preformed based on the consent of the patient’s family. These processes were quite complex, so we did not include these patients in this study.
BP management in patients with sICH is always a heated topic among neuroscientists. A lot of observational studies showed that the elevated BP, especially the SBP, might be related to the enlargement of hematoma or recurrent hemorrhage, which would lead to neurological deterioration and poor outcome directly22,25,31). Some further well-designed randomized controlled trials showed that intensive BP management, which means to lower the SBP below 140 mm rapidly, is safe and might be effective among patients with spontaneous intracarebral hemorrhage1–3,10). In addition to the level of BP, the variability of SBP is also an important risk factor of neurological deterioration and unfavorable outcome14,28). However, the present study did not find any association between BP and rehemorrhage in postoperative patients with sICH. Further studies on blood management for postoperative patients with sICH were still needed.
Previous studies confirmed that the diabetes mellitus is a risk factor of both ischemic and hemorrhagic stroke21). Further studies showed admission level of glucose was related to early death and poor outcomes in patients with sICH27,29). In addition, the diabetes mellitus is also associated with cerebral microbleeds and recurrent intracerebral hemorrhage8,32). Results of this study indicated that diabetes mellitus was a risk factor of rehemorrhage after surgery. To our best of knowledge, there was no prior study found an association between hematoma enlargement or rehemorrhage and diabetes. Atherosclerosis of small arterials and changing of microcirculation caused by chronic diabetes mellitus might be the reasons.
Results of the present study showed that the midline shift is another independent risk factor of rehemorrhage in postoperative patients with sICH. Midline shift is an important neuroimaging parameter, which could predict outcome independently in patients with sICH13). It was reported that midline shift was associated with ICP caused by hematoma and peri-hematoma edema9). Patients with higher ICP would have a more serious ischemia reperfusion injury which might be associated with rehemorrhage after hematoma evacuation.
In the present study, even though 2 independent predictors of rehemorrhage were revealed, we are unable to identify any method to prevent rehemorrhage after surgery. However, in consideration of the results, patients with diabetes mellitus or greater midline shift should be carefully observed after surgery because of higher risk of rehemorrhage. The BP should be kept low and stable in postoperative patients with sICH, although level and variability of SBPs after surgery were not independent factors associated with rehemorrhage. In addition, coagulation function should be monitored in posteroperative patients.
There are some limitations of this study. First, this study was a retrospective study and there might be a confounding bias potentially. The sample size of this study was not large enough to provide robust evidence for clinical practice. Second, Postoperative reexamine of coagulation functions and level of alcohol intake were not analyzed in this study because of incomplete data.
CONCLUSION
In conclusion, our study identified that diabetes mellitus and midline shift were possibly associated with rehemorrhage in postoperative patients with sICH. Finding of the present study might help doctors to identify the patients with higher risk of rehemorrhage.
PATIENT CONSENT
The patient provided written informed consent for the publication and the use of their images.
Acknowledgements
The biggest acknowledgement goes to the patients who have taken participate in this study and also to everyone who makes a contribution to make this research possible.