Nonprogressive Unilateral Intracranial Arteriopathy in Children with Arterial Ischemic Stroke
Article information
Abstract
The nonprogressive unilateral intracranial arteriopathy known as transient (focal) cerebral arteriopathy is not a well-recognized arteriopathy among practitioners of Korea and Japan, although it cannot be easily differentiated from early moyamoya disease. This review summarizes the nomenclature, pathophysiology, diagnostic evaluation, clinico-radiological features, and management of nonprogressive (reversible or stable) unilateral arteriopathy based on the relevant literature and our own experiences. Nonprogressive unilateral arteriopathy should be strongly suspected in children presenting with basal ganglia infarction and arterial beading. The early identification of patients likely to have nonprogressive or progressive arteriopathy would ensure proper management and guide further research for secondary stroke prevention.
INTRODUCTION
Traditional risk factors for arterial ischemic stroke (AIS), such as smoking, hypertension, hypercholesterolemia, and diabetes mellitus, are uncommon in children. Instead, childhood AIS risk factors include arteriopathy, congenital heart disease, sickle cell disease, infection, and hematologic abnormalities. Although pediatric moyamoya disease is considered as a major cause of childhood AIS in East Asia, the nonprogressive, often reversible, unilateral intracranial arteriopathy known as transient cerebral arteriopathy (TCA) or focal cerebral arteriopathy (FCA) has been recognized as a leading cause of childhood AIS worldwidely17). The pathophysiology of TCA is still poorly understood, but is thought to be a self-limited inflammatory process, generally extending for 1 to 3 months25). The term "transient" may be misleading because the arteriopathy may persist as a stable form on long-term follow-up imaging. In the initial worsening stage, TCA may not be distinguished from unilateral moyamoya disease or other progressive arteriopathies such as primary angiitis (vasculitis) of the central nervous system in children (cPACNS)4725). However, TCA is not a well-recognized arteriopathy in East Asian countries where moyamoya disease is the principal cause of childhood AIS.
Recently, the authors retrospectively reviewed the records of 74 Korean children and adolescents (6 months to 17 years of age) who presented with AIS and intracranial arteriopathy to identify 29 patients with unilateral large-artery arteriopathy mainly in the anterior circulation32). Patients with only transient ischemic attacks were not eligible for the study and 25 patients who fulfilled the following inclusion criteria were analyzed to determine the angiographic course and outcome : 1) repeated vascular imaging at least twice and 2) absence of thrombotic disorders or cardiac diseases. Interestingly, the course of unilateral arteriopathy was usually reversible (68%) rather than progressive (20%) or stable (12%). This review summarizes the nomenclature, pathophysiology, diagnostic evaluation, clinico-radiological features, and management of nonprogressive (reversible or stable) unilateral arteriopathy based on the relevant literature and our own experiences.
NOMENCLATURE
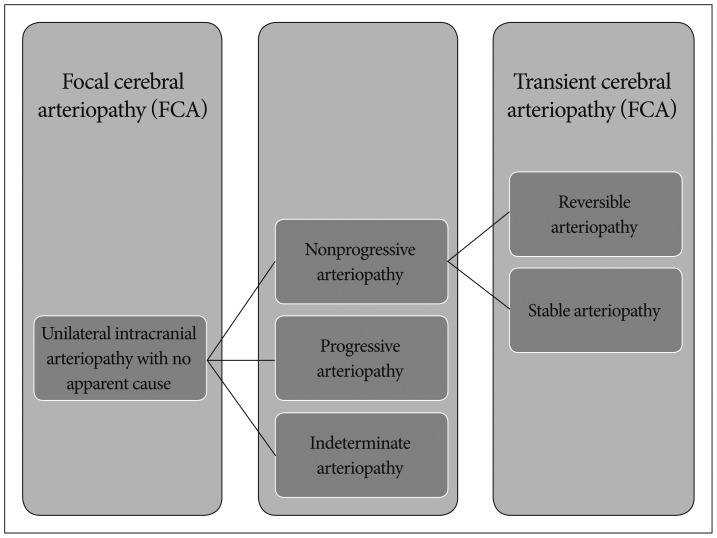
Arteriopathy is an important cause of childhood AIS. Over half (277 of 525; 53%) of subjects under 19 years of age enrolled in the International Pediatric Stroke Study (IPSS) had an arteriopathy119). Although data from East Asian countries is limited, FCA supplying the infarct territory was the most common type of arteriopathy observed (69 of 277 children; 25%), followed by moyamoya disease/syndrome (61 of 277 children; 22%)1). In the IPSS, the term FCA was defined as arterial stenosis on vascular imaging not otherwise classified as dissection, moyamoya, sickle cell arteriopathy, postvaricella arteriopathy, vasculitis, or other specific diagnoses (such as post-irradiation arteriopathy)1). Thus, FCA is a newly-coined descriptor for unilateral intracranial arteriopathy with no apparent cause, and it could be applied at baseline regardless of changes in arteriopathy over time (Fig. 1)6).
Similarly, unilateral anterior circulation arteriopathy was the most common cause of childhood AIS (128 of 372 children; 34%) in a multicenter European study7). Moreover, 74 of 79 (93.7%) previously healthy children with available imaging follow-up were found to have nonprogressive unilateral arteriopathy [reversible TCA in 50 (63.3%) patients and stable TCA in 24 (30.4%) patients]7). The definition of TCA includes two primary criteria : 1) unilateral steno-occlusion involving the distal part of internal carotid artery (ICA) and the initial segments and branches of the anterior cerebral artery (ACA) and middle cerebral artery (MCA) and 2) occasional worsening of the arteriopathy <3 months but non-progression >6 months after AIS2526). Because the diagnosis of TCA depends on the time course of arteriopathy on repeated vascular imaging, cases that might eventually meet the criteria for TCA would be classified as FCA at baseline (Fig. 1)6).
In the pediatric rheumatology literature, intracranial arteriopathy not attributable to other causes has been labeled cPACNS. These can be further classified into progressive or nonprogressive forms based on arteriopathy progression >3 months4). Progressive cPACNS is characterized by initial neurocognitive dysfunction, bilateral multifocal and gray matter lesions, angiographic patterns of multifocal stenoses, bilateral involvement, and distal vessel stenoses. The clinico-radiological features and course of nonprogressive cPACNS are very similar to those of TCA, suggesting that these two conditions are likely the same illness415).
The classification system of childhood AIS is currently in development, and the best available system is the Childhood AIS Standardized Classification and Diagnostic Evaluation (CASCADE) criteria based on the anatomic (acute primary classification) and temporal features (chronic primary classification)6). According to the acute primary classification criteria, childhood AIS can be categorized into seven types : small vessel arteriopathy of childhood, unilateral focal cerebral arteriopathy of childhood (FCA), bilateral cerebral arteriopathy of childhood, aortic/cervical arteriopathy, cardio-embolic, other, and multifactorial. The courses of FCA as well as bilateral cerebral arteriopathy and aortic/cervical arteriopathy are ultimately determined as progressive, stable, reversible, or indeterminate (incomplete follow-up) based on repeated vascular imaging at 3 to 6 months or more (chronic primary classification)6).
PATHOPHYSIOLOGY
TCA is a provisional diagnosis that does not imply an underlying mechanism25). Indeed, it may represent the end result of a variety of underlying pathophysiological mechanisms producing the same angiographic appearance, including inflammation, infection, and dissection110). Historically, varicella-zoster virus has received particular attention because of its ability to invade vessel walls, possibly by direct spread from adjacent cranial nerves18). Multiple case reports have described arteriopathic childhood AIS within weeks to months of chicken pox (primary varicella zoster virus infection); rarely, herpes zoster (secondary varicella zoster virus infection); varicella zoster IgG antibodies or DNA has been detected in the cerebrospinal fluid91622). In a fatal case of progressive postvaricella stroke, varicella zoster virus antigen-positive arteritis of the MCA was demonstrated pathologically5). The prevalence of chicken pox in the year before the AIS was higher (31%) than the annual prevalence of chicken pox reported in the regional population (9%)3).
Postvaricella arteriopathy is defined by the same diagnostic criteria as for TCA in a patient with a history of varicella infection within 12 months before AIS26). Varicella zoster virus infection is present in a many (44-64%) of TCA patients71227). However, postvaricella arteriopathy is virtually indistinguishable from TCA in terms of clinico-radiological characteristics and outcomes7132627). Other infectious agents, such as enterovirus and Borrelia, are also associated with TCA25). Recent upper respiratory infection was implicated as a predictor of FCA in the IPSS study. When 69 children with FCA were compared with 456 children without FCA (either no arteriopathy or a defined arteriopathy), the only significant univariate predictor of FCA was recent upper respiratory infection (OR, 2.81; 95% CI, 1.28 to 6.12; p=0.003)1).
The role of infection in vascular injury can include direct invasion by the pathogen, promotion of vascular cell-wall infiltration by inflammatory cells, stimulation of vascular smooth muscle cell proliferation, or activation of prothrombotic states229). Therefore, infection is both a differential diagnosis (e.g., direct infection of cerebral arteries) and a potential trigger for inflammatory cerebral arteriopathy (e.g., parainfectious or postinfectious)20). Considering various infectious agents and cases without a definite history of infection, the likely mechanism would be a self-limited inflammatory process of unknown causes inducing arterial wall inflammation and thrombosis.
Recently, the Vascular Effects of Infection in Pediatric Stroke (VIPS) Study, which exploited the existing infrastructure of the IPSS, was designed to test the hypothesis that infection predisposes children to arteriopathy, arterial ischemic stroke, and recurrence14). The VIPS study enrolled 355 cases of AIS between January 2010 and March 2014. Of these, 127 cases (36%) of definite arteriopathy were present30). However, 10% of all cases could not be definitively classified as having/not having arteriopathy, even with central review by a panel of pediatric stroke investigators30). Significant variability in patient evaluation was the main limitation of the VIPS study. Only half of the cases had cervical imaging, only 41% had follow-up imaging, and only 14% had catheter angiography30). The investigators are currently planning further work to determine predictors of arteriopathy subtypes, with the aim of developing simple algorithms for differential diagnosis.
DIAGNOSTIC EVALUATION
All patients with AIS should undergo standard blood and cardiac investigations to rule out other possible causes of AIS24). Standard blood investigations include a complete blood count, chemistry profile, lipid profile, inflammatory markers (erythrocyte sedimentation rate, C-reactive protein, and, if necessary, serum autoantibodies), and thrombotic disorder profile (fibrinogen, thrombin time, D-dimer, antithrombin III, plasminogen, plasminogen activator inhibitor, protein C, protein S, activated protein C resistance, dilute Russel viper venom time, lupus anticoagulant, and homocysteine).
Cardiac investigations include chest X-ray, electrocardiography, and transthoracic echocardiography with contrast. The presence of cardiac disease including patent foramen ovale should be evaluated because reversible arteriopathy in these patients might have been attributable to thromboembolic steno-occlusion and later recanalization. In cases with isolated patent foramen ovale, the direction and size of shunting also need to be documented, although cardiac disease can be coincident with arteriopathy. Cerebrospinal fluid examination may be normal or non-specific (mild to moderate lymphocytosis) in cases with TCA, although varicella zoster IgG antibodies can be detected in patients with a prior history of varicella zoster infection23).
Magnetic resonance angiography (MRA) is a preferred vascular imaging modality at the time of AIS (at least within 1 to 2 days). This should include cervical arteries and may be performed with contrast enhancement. Time-of-flight MRA is not less sensitive than contrast-enhanced MRA in detecting stenosis in the major intracranial artery and can be consistently used for determining the course of arteriopathy, especially in younger children. Cases showing no or only mild abnormalities on the initial MRA may need repeated MRA after a few days. This is because TCA may initially be present with only mild abnormalities (irregularity) and progress to severe stenosis within days30). Unilateral arteriopathy on the initial MRA needs to be reevaluated by catheter angiography (digital subtraction angiography, DSA). In a study comparing time-of-flight MRA at 3T with DSA, the positive predictive value of MRA in detecting >50% intracranial stenosis was 75 to 79%, whereas the negative predictive value of MRA was 95 to 97%11).
The evolution of unilateral arteriopathy is determined as improved, worsened, or unchanged by comparing the findings (apparent luminal diameter as well as the longitudinal extent of arteriopathy and distal filling) of identical angiographic techniques (MRA or DSA). The overall course of unilateral arteriopathy is subsequently determined as reversible, progressive, or stable (indeterminate in cases with incomplete follow-up)67). Reversible unilateral arteriopathy is defined as an arteriopathy showing any improvement including normalization (Fig. 2). Initial (transient) worsening within 6 months (mostly within 1 month) is also considered indicative of reversible arteriopathy if the subsequent imaging demonstrates any improvement. Progressive unilateral arteriopathy is characterized by continued worsening of arteriopathy beyond 6 months or progression to bilateral arteriopathy.
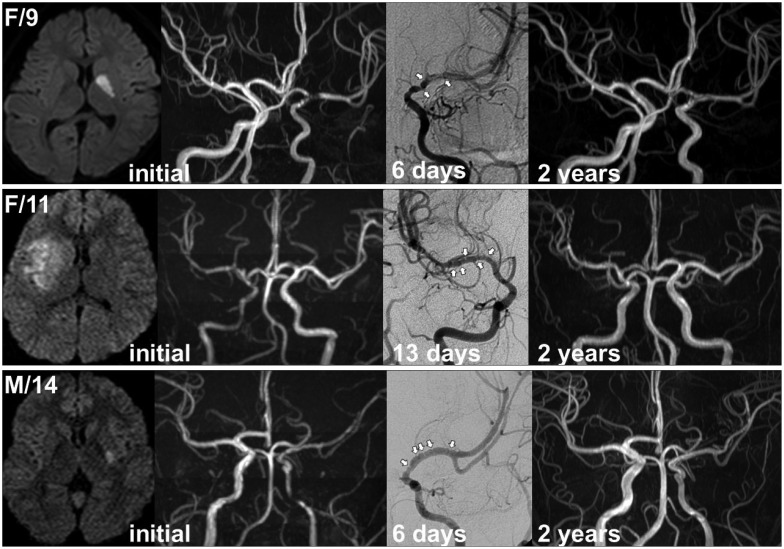
Examples of reversible arteriopathy. Note the infarction involving the basal ganglia and arterial beading (white arrows) on digital subtraction angiography performed within 2 weeks.
Stable unilateral arteriopathy is defined as an arteriopathy that remains unchanged beyond 6 months or has been stabilized after initial worsening (worsening of arterial lesions within the first 6 months but no change thereafter)67). Notably, cases that fulfill the definition of reversible or stable arteriopathy vary significantly depending on the frequency and interval of repeated vascular imaging. In the absence of repeated vascular imaging at 1 and 3 months, initial worsening and early improvement within 3 months may go undetected. Additionally, considering that the progression of unilateral moyamoya disease may be delayed for years31), long-term follow-up (e.g., >3 years) is required to confirm the diagnosis of stable or stabilized arteriopathy (Fig. 3).
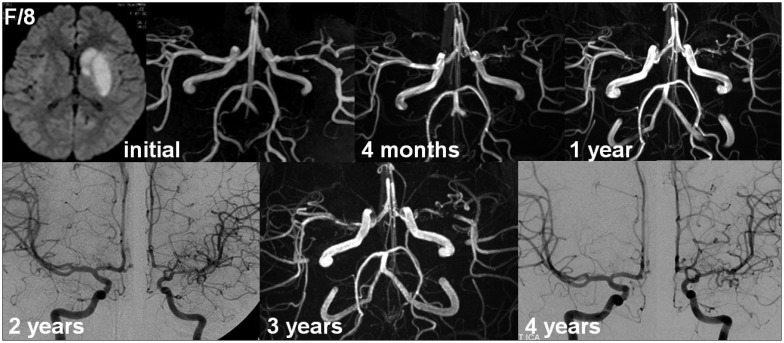
Example of stable arteriopathy. The left middle cerebral artery (MCA) stenosis worsened on magnetic resonance angiography (MRA) at 4 months but remained unchanged thereafter. Although contralateral MCA stenosis was suggested by MRA at 1 year based on the decreased luminal diameter, subsequent angiography confirmed no definite contralateral progression.
Arteriopathy worsening on follow-up MRA may be attributable to the imaging artifact of time-of-flight MRA and must be reevaluated by repeated DSA or subsequent MRA (with or without contrast-enhanced MRA). However, repeated DSA exposes children to a substantial amount of radiation and requires repeated general anesthesia in younger children. Moreover, the initial DSA performed in the initial worsening stage of reversible arteriopathy may demonstrate apparently worsened arteriopathy as compared to the initial MRA findings. Thus, repeated DSA may be reserved until progressive arteriopathy is ascertained by subsequent MRA (further worsening thereafter). The improved arteriopathy on follow-up MRA may be followed by subsequent MRA alone considering the high sensitivity of time-of-flight MRA in detecting major artery stenosis. But, improvement on MRA should be cautiously determined based on increased flow signals from the affected artery as compared to unchanged flow signals from unaffected arteries.
CLINICO-RADIOLOGICAL FEATURES
In children, the clinical recognition of stroke is often difficult, leading to a considerable time delay between the onset of symptoms and the first encounter with a healthcare provider7). Hemiplegia is the most common presentation of childhood AIS irrespective of stroke etiologies. The mode of onset may differ in children with AIS and nonprogressive unilateral arteriopathy. In a retrospective study analyzing 30 children with TCA (including postvaricella arteriopathy), 23 (77%) children exhibited nonabrupt onset of neurological symptoms defined as a course that was progressing (n=6, gradual progression of neurological symptoms reaching maximum severity >30 minutes), stuttering (n=8, fluctuating neurological symptoms), or recurring (n=6, recurrent episodes of transient neurological deficits before the index stroke)8).
Most patients with nonprogressive unilateral arteriopathy present with basal ganglia infarction with or without MCA territory cortical infarction732). More specifically, the pattern of infarction in our 17 patients with reversible arteriopathy was classified as isolated basal ganglia infarction (n=9, 53%), basal ganglia and distal MCA infarction (n=6, 35%), and isolated distal MCA infarction (n=2, 12%)32). In contrast, the pattern of infarction in our 5 patients with progressive arteriopathy was classified as isolated basal ganglia infarction (n=1, 20%), isolated distal MCA infarction (n=1), distal MCA and ACA infarction (n=1), and borderzone infarction (n=2, 40%)32). Meanwhile, children and adolescents with unilateral moyamoya disease almost always present with transient ischemic attacks rather than ischemic or hemorrhagic stroke31).
The arteriopathy affects the distal part of ICA and the initial segments and branches of ACA and MCA. Ipsilateral posterior cerebral artery involvement is rare, but may occur in select patients depending on the variations of the posterior communicating artery2832). An interesting finding of nonprogressive unilateral arteriopathy is arterial beading (alternating short segments of stenosis, also known as banding) on the initial DSA performed within 2 weeks, as observed in 83% of our patients with reversible arteriopathy (Fig. 2)32). Although beading may be considered nonspecific7), it has been usually associated with reversible conditions and diseases like TCA, reversible cerebral vasoconstriction syndrome, and nonprogressive cPACNS417). The absence of abnormal collateral networks such as moyamoya vessels would be another distinguishing feature of nonprogressive unilateral arteriopathy although two of our 5 patients with progressive arteriopathy did not exhibite abnormal collateral networks on the initial angiography.
Notably, children with AIS and nonprogressive unilateral arteriopathy may suffer from an additional infarction in the initial worsening stage. Previously, we classified the overall course of unilateral arteriopathy as reversible in 17 of the 25 patients (68%) during the mean radiological follow-up of 37 months32). Nine patients exhibited initial worsening of the arteriopathy that almost always occurred within 1 month. Two of these nine patients experienced clinical worsening at 6 days with an additional infarction (stroke progression). However, the worsened arteriopathy began to improve within 3 months and continued to improve even after a few years. Eventually, the arteriopathy nearly normalized in 12 of the 17 patients. None of the 17 patients exhibited worsening after improvement.
Unlike the high prevalence of stable arteriopathy in the European multicenter study (30%)7), only 12% (3 of 25) of our patients were regarded as having stable unilateral arteriopathy that remained unchanged (n=1) or stable (stabilized) after initial worsening (n=2)32). Moreover, while only 20% of the patients with reversible arteriopathy demonstrated initial worsening (median interval of 3.5 months) in the European study, 53% of our patients exhibited initial worsening that almost always occurred within the first month after AIS. These differences would best be explained by a longer radiological follow-up and more frequent vascular imaging in our previous study.
Considering the typical time course of initial worsening within 1 month and early improvement within 3 months, repeated vascular imaging at 1, 3, and 6 to 12 months would be necessary to differentiate nonprogressive arteriopathy clearly from progressive arteriopathy. In the absence of vascular imaging at 1 or 3 months, the worsened arteriopathy at 6 months might be either progressive arteriopathy or residual but improving arteriopathy after maximal worsening. Similarly, if the arteriopathy seemed unchanged at 6 months, the course of arteriopathy might be truly stable, improving after maximal worsening, or even slowly progressive (beyond detection).
MANAGEMENT
Despite the fact that stroke progression may occur in the initial worsening stage, there is very limited information concerning acute treatment. Given the presumed mechanism of arterial wall inflammation and thrombosis, the use of immunosuppressive drugs including steroids and antithrombotic drugs like heparin may be considered in the acute phase4721). Two of our 17 patients with reversible arteriopathy were managed with high-dose steroid therapy consisting of methylprednisolone 20-25 mg/kg for 5 to 7 days. Despite the worsened arteriopathy during the acute phase, both patients did not experience clinical deterioration and no additional infarction was demonstrated on magnetic resonance imaging. Among the other 15 patients, stroke progression occurred in two patients 6 days after the index stroke. At the time of stroke progression, one patient continued to take aspirin and the other patient began to take aspirin for 2 days after 4 days of heparinization. The possible benefits versus risks of immunosuppressive therapy, combined with antithrombotic and/or antiviral therapy, need to be determined in future studies.
Although the inflammatory process involved in nonprogressive unilateral arteriopathy is considered to be self-limiting, patients often have residual arteriopathy for a considerable time or may have chronic stabilized arteriopathy. Therefore, many patients are treated with antiplatelet medication to prevent recurrent stroke but for an undetermined duration of treatment471213). Because of the nonabrupt onset of neurological symptoms and stroke progression in the initial worsening period, recurrent stroke can be defined as any neurological deterioration accompanied by new areas of infarction >1 to 2 weeks after the index stroke713).
In the European multicenter study, the reported rate of recurrent stroke after 1 week was 10% (5 of 50 patients) among patients with reversible arteriopathy, whereas it was 21% (5 of 24 patients) among patients with stable (stabilized) arteriopathy7). The rate of recurrent stroke did not differ between patients with normalized arteriopathy (2 of 17 patients; 12%) and patients with improving but residual arteriopathy (3 of 33 patients; 9%)7). Antiplatelet medication was prescribed in 78% of patients with nonprogressive arteriopathy, but no definite relationship between recurrent stroke and antiplatelet therapy was observed7). Meanwhile, two other studies reported no cases of recurrent stroke after 2 weeks of the index stroke with prolonged aspirin treatment1213).
In our previous study, long-term antiplatelet medication (mostly aspirin) was prescribed to 12 (60%) of the 20 patients with nonprogressive (reversible and stable) arteriopathy. Recurrent stroke developed in only one patient (5%) at 14 months after the index stroke. However, this patient was taking aspirin at that time and recurrent stroke occurred after recent upper respiratory infection with no apparent change in the improving but residual MCA stenosis. In contrast, none of the eight patients managed without antiplatelet medication experienced recurrent stroke during the mean follow-up period of 39 months. These observations raise doubt about the efficacy of long-term antiplatelet therapy in children with AIS and nonprogressive unilateral arteriopathy. Randomized clinical trials are needed to determine appropriate treatment strategies.
SUMMARY
Nonprogressive unilateral arteriopathy should be strongly suspected in children and adolescents with AIS involving the basal ganglia and arterial beading on DSA within 2 weeks of AIS. The absence of moyamoya vessels and rarity of posterior cerebral artery involvement may offer additional clues. A high index of suspicion would reveal more cases, since some patients may present with only equivocal stenosis on the initial MRA. Considering the typical time course of initial worsening within 1 month and early improvement within 3 months of AIS, repeated vascular imaging at 1, 3, and 6 to 12 months is necessary to differentiate nonprogressive arteriopathy clearly from progressive arteriopathy. However, stroke progression may occur in the initial worsening stage. The early identification of patients likely to have nonprogressive or progressive arteriopathy would ensure proper management and guide further research for secondary stroke prevention. Dedicated vessel wall imaging and investigations for inflammatory causes of AIS may help to establish the correct diagnosis and treatment.