Kim, Cho, Chang, and Youn: Clinical Outcome of Treatment for Patients with Giant Cell Tumor in Spine
Abstract
Objective
The treatment of giant cell tumor (GCT) is mainly performed surgically. However, GCT in spine seems difficult to treat because of the limited surgical accessibility and proximity. In this report, we analyzed the outcome of GCT treatment in spine.
Methods
Between 2000 and 2012, 19 patients received treatment for GCT in spine. Median age at their first diagnosis was 31 years, 10 patients were male, and 9 female. Fourteen tumors were located in the sacrum, 1 in cervical, 1 in thoracic and 3 in lumbar spine. As primary treatment, gross total removal (GTR) was done in 6 patients, and subtotal removal (STR) in 13 patients. Radiation therapy (RT) as an adjuvant therapy was performed in 2 cases in GTR group and 10 cases in STR group.
Results
During the follow-up, 7 patients had local recurrence (36.8%). The average period until recurrence after primary treatment was 14 months. No recurrence was detected in GTR group. Recurrence was noted in 7 out of 13 patients who underwent STR. These differences were statistically significant (p=0.024). A median of recurrence free period (RFP) was 84 months. Also average RFP of the RT group was 112 months, and non-RT group was 65 months. These differences were statistically significant (p=0.041).
Conclusion
Treatment of choice for GCT in spine is a complete removal of tumor without neurological deficits. In case of incomplete removal, radiation therapy may be a useful adjuvant treatment modality.
Key Words: Giant cell tumor ┬Ę Recurrence ┬Ę Radiotherapy.
INTRODUCTION
Giant cell tumor (GCT) accounts for about 7 to 10% of all cases of primary spinal tumors. Patients with GCT are usually diagnosed in the third or fourth decade of life, and there is a slight female preponderance 15,2325). GCT usually occurs in the metaepiphyseal ends of the long bone, and it rarely occurs in the spine 2,611). Savini et al. 28) reported that only 2.9% of all GCT incidences occur in the spine, and Goldenberg et al. 14) reported that spine involvement is 1.3% in a study of 218 cases of GCT. Moreover Dahlin 10) noted about 4.2% of spine GCT occurrence. A research conducted by Mayo Clinic reported incidence of up to 6.5% of GCT in the spine 27). GCT in the spine usually involves sacrum, but it also occurs in other parts of the spine. GCT is classified as benign tumor histopathologically, but it had a locally aggressive tendency. And, if it is incompletely excised, it shows a high recurrence rate. There is a reason for a relatively worse prognosis in GCT patients, compared to other benign primary spinal tumor patients 7,826). Many studies reported from 10 to 40% local recurrence rate after spinal GCT treatment, depending on surgical protocols 4,614,1821). In case of recurrence, excision is conducted when another operation is possible. If re-operation is considered difficult, radiation therapy (RT) is introduced. Generally, treatment of choice for GCT is wide en bloc excision. Many studies recommend the treatment for GCT in spine as well 4,1416,1821). However, due to the characteristics of spine, there is a possibility of neural or vascular injury, which will result in difficult wide en bloc excision 10). Radiation therapy may be an option in case of incomplete excision or recurrence. However, RT has remained controversial. Some authors reported that RT is not effective in local tumor control and has a risk of malignant transformation of GCT 15,2527,28). Recent studies reported chemotherapy as effective in case of recurrence or when there are complications 1,59,1329). The objective of this study is to obtain the results of treatment GCT in the spine. In addition, we analyzed whether local recurrence is influenced by surgical protocols, or RT.
MATERIALS AND METHODS
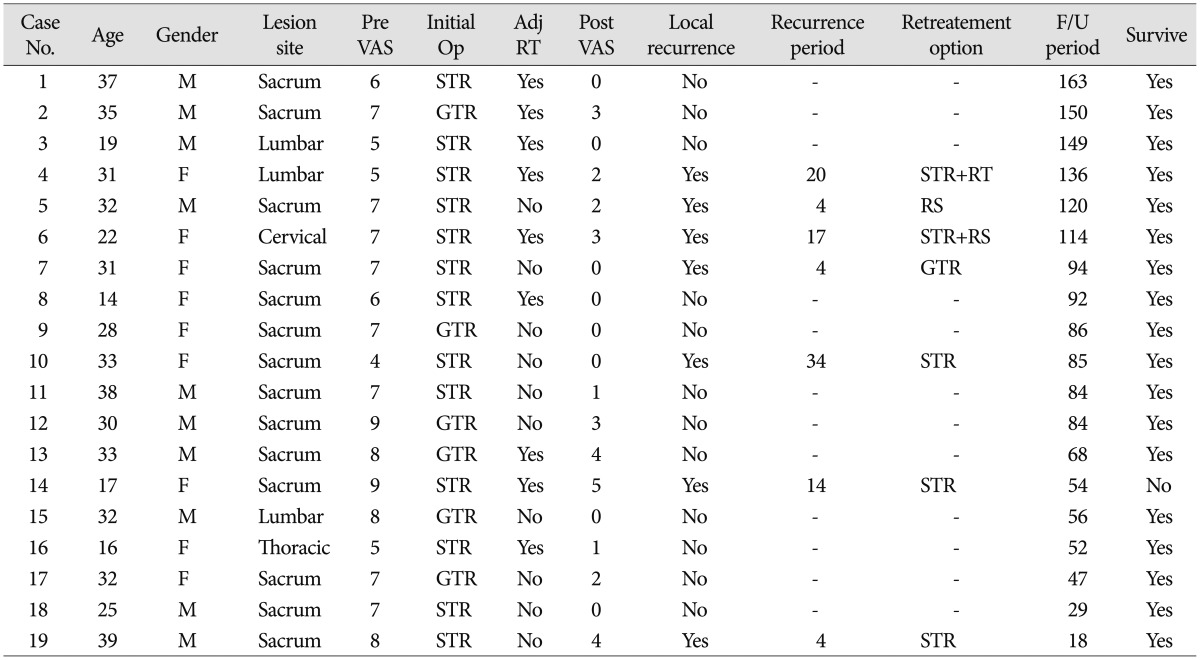
The subjects of our study were 242 patients treated for GCT from 2000 to 2012. Patients with involved GCT spine were 19 cases, and incidence rate was 7.9%. Their medical records and radiological images were retrospectively reviewed. Table 1 demonstrates an overview of the patients. The median age at their first diagnosis was 31 years (range, 14 to 39 years). Ten patients were male, and 9 were female. Fourteen tumors were located in the sacrum, one in cervical, one in thoracic, and three in lumbar spine. All the lesions were single and localized at the original spine except one, which originated in the sacrum extending into the coccyx. The median follow-up period was 92 months (range, 18 to 163 months). Main symptom of the patients with GCT was pain, and the average visual analogue scale (VAS) was 7 (range, 5 to 9). Only one out of 19 showed motor weakness initially caused by spinal cord compression at thoracic spine. For initial treatment, 6 out of 19 patients underwent gross total removal (GTR), and 13 patients underwent subtotal removal (STR). Wide en bloc excision was not attempted. In case of GTR, all the involved structures were removed completely. STR was defined as cases in which remnant tumor remained macroscopically. Adjuvant RT was performed in 12 cases, 2 cases in GTR group and 10 cases in STR group.
We analyzed the recurrence rate and recurrence free period (RFP) for GCT in the spine after the treatment. In addition, we analyzed the difference of the recurrence rate according to the surgical protocol and RT. We did not consider the effect of the treatment for the patients with recurrence after the initial treatment.
Recurrence analysis was performed by using log-rank test. Recurrence free curves were created by using Kaplan-Meier life-table analysis. Chi-square tests were used for categorical variables, and Student's t-tests were used for continuous and ordinal variables, as appropriately. A p value 0.05 (two-tailed) was considered significant. The data were compiled and analyzed with the software package SPSS, version 18.0 (SPSS Inc., Chicago, IL, USA).
RESULTS
During the follow-up period, 7 out of 19 patients had local recurrence, at 36.8% rate. One patient among them showed pulmonary metastasis. An average recurrence free period was 14 months (range, 4 to 34 months) for the patients with recurrence. Median recurrence free period of all patients was 84 months (95% CI, from 4 to 163 months) ( Fig. 1). For local recurrence rate depending on surgical protocols, GTR group did not have any recurring patients, while 7 out of 13 patients who received STR had local recurrence ( Table 2), which is statistically significant ( p=0.024). This implies that GTR is the most important method of treatment for controlling the tumor. Regardless of surgical protocols, 12 out of 19 patients received adjuvant RT. As for the local control effect of radiation therapy, the average recurrence free period of the group that underwent radiation therapy was 112 months (95% CI, from 74 to 150 months), which was longer than that of the group without radiation therapy, for 65 months (95% CI, from 47 to 83 months) ( Fig. 2). Because there was a statistically significant difference between RT group and non-RT group ( p=0.041), we considered that radiation may be effective in local control of tumors. Seven out of 13 patients in STR group had recurrence. Four patients underwent re-operation, 2 patients underwent re-operation and RT, and 1 patient underwent radiosurgery. GTR was possible in only one out of 6 patients who underwent re-operation.
All patients reported relieved pain after the treatment according to VAS (range, 0 to 5). The average VAS was 2, compared to the initial average VAS of 7. Nine out of 19 patients were pain-free during the follow-up period. The patient with motor weakness showed incomplete recovery after surgery, although the improvement was not functional and bladder dysfunction remained.
Neurological complication after surgery occurred in 3 out of 19 patients, including bladder or bowel dysfunctions. All the patients who experienced complications had GCT in the sacrum. Until the final follow-up period, 18 out of 19 patients remained alive without recurrence, and 1 patient with pulmonary metastasis expired due to pulmonary complication.
Case presentation
We present 2 cases of treatment.
Case 1
Female 31-year-old patient visited our hospital with pain in buttock that lasted for 4 months. Magnetic resonance (MR) image showed a huge enhancing mass involving the sacrum ( Fig. 3A). The patient underwent STR, and the pathologic examination revealed to be GCT. On Computed tomography scan (CT) after the operation showed remnant tumor on the ventral side of S2 spinal canal ( Fig. 3B). Adjuvant treatment was not performed. Four months after the initial operation, MR image showed the progression of the tumor extending to the left side ( Fig. 3C). The patient underwent GTR. Ninety-four months after re-operation, the tumor did not recur ( Fig. 3D).
Case 2
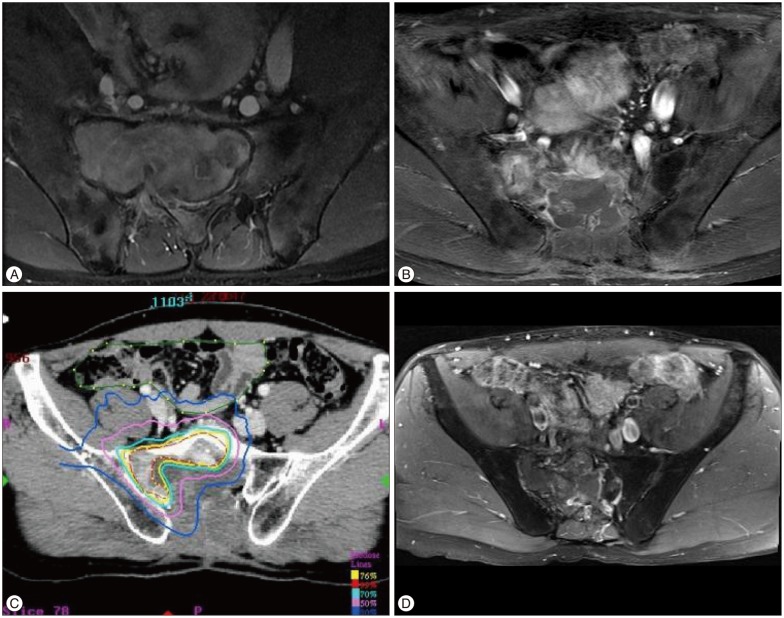
33-year-old male patient complained lower back pain and radiating pain into both legs for 4 months. MR image showed a huge enhancing mass involving the entire sacrum ( Fig. 4A). STR was performed ( Fig. 4B). Cyberknife® (Accuray, Inc., Sunnyvale, CA, USA) radiosurgery was done to the remnant tumor located in front of the sacrum ( Fig. 4C). The patient's pain was relieved and he has been stable for 68 months ( Fig. 4D).
DISCUSSION
The GCT usually involves the end of long bone as femur (23%), tibia (21%), and fibula (5%) around knee, and a few other arise from the spine (range, 3% to 7%) 24). Spinal GCT usually arises in the sacrum 7,8). It is reported that there is a slight female preponderance in spinal GCT 15,2325). In our study, the incidence of spinal GCT was 7.9%, and the male to female ratio is 1 to 0.9. Treatment of choice for GCT patients is en bloc wide excision. Total excision is also considered when recurrence occurred 4,614,1821). However, GCT in the spine is not easy to remove totally because of lack of accessibility and adjacent important neural and vascular structures 10). In case of incomplete excision, RT can be considered, but RT for GCT is still controversial 15,1625,2728). Wide or marginal excision of the tumor or en bloc resections may yield in a lower recurrence rate. Liljenqvist et al. 20) stated on malignant tumors of the spine that en bloc spondylectomy enables wide or marginal resection in most cases with acceptable morbidity. Compared to a degree of resection, the textbook showed average of 50% (from 35 to 70%) in curettage alone and from 10 to 15% recurrence rate in en bloc resection 24). Campanacci et al. 6) reported a recurrence rate of 27% after intralesional curettage versus rates of 8% and 0% after marginal or wide resection, respectively. Fidler reported successful result of only one recurrence in 9 consecutive patients who underwent en bloc resection 12). In a larger series, 10 of 32 patients who had intralesional curettage recurred within 16 months postoperatively and none of 2 patients with en bloc resection recurred 15). Regardless of its surgical resection, 25 to 28% of recurrence rate showed in other series 15,2728). According to Kim et al. 16), 32.4% of recurrence rate in curettage and 11.1% of rate in en bloc resection (overall 27.1%) was 16). In our study, GTR was defined as cases in which all the involved structures were removed completely. STR was defined as cases in which remnant tumor remained macroscopically. Average recurrence rate of all patients was 36.8%, with 0% recurrence rate in GTR group, and 53.8% in STR group. The surgical protocols showed a statistically significant difference ( p=0.024), implying gross total resection as more desirable. We performed GTR rather than en bloc wide excision but there was no recurrence case during the median follow up period 92 months. In addition, 14 out of 19 cases located in the sacrum. En bloc resection of the sacrum is much more complicated than that of the thoracolumbar spine. Most report about en bloc spondylectomy for GCT mentioned above localized in the thoracolumbar spine. In case of the recurrence (case 4), the patient received additional surgery and RT. She survives longer than 136 months in spite of the recurrence. Considering the morbidity of en bloc wide excision in spinal GCT, GRT is not worse than en bloc wide excision. In the early 2000s, STR and RT was used to treat spinal GCT in our hospital, which was the reason for high recurrence rate. Later, STR was considered not effective for local control of GCT, which led to a wider use of GTR. The efficacy of RT is still controversial. RT may be ineffective for tumor control or may cause malignant sarcomatous transformation 15,1625,2728). However, recent studies insisted that RT is not only a useful adjuvant treatment modality after incomplete removal of tumors but also an effective therapy as a sole treatment of GCT of the spine 15,2527,28). Hart et al. 15) reported that 8 out of 36 patients received RT as an initial treatment before surgery, no case except one recurred. One GCT progressed to osteosarcoma. Two other patients who had RT as a sole treatment experienced no recurrence. Four patients had RT after recurrence, and only 1 patient experienced re-recurrence. Kim et al. 16) reported that regardless of surgical protocols, 29 out of 96 cases had received RT and only 3 cases (10.3%) of recurrence occurred. In 67 cases without RT, 23 cases (34.3%) of recurrence occurred, which is significant statistically ( p=0.031) 16). In our study, although recurrence rate was 42% after RT, average recurrence free period was 112 months for RT group, and 65 months for non-RT group, with statistical significance ( p=0.041). There were no cases of malignant transformation. Even after recurrence, additional RT or radiosurgery after re-operation yielded good results. RT seems to be a good treatment modality for delaying recurrence and locally controlling the tumor. Distant metastasis of GCT is reported to be about 2 to 9% 17). In our study, only 1 out of 19 patients (5%) had pulmonary metastasis. Bertoni et al. 3) mentioned that RT or chemotherapy may be useful to treat pulmonary metastasis of GCT. Other adjuvant therapies, such as a cryotherapy that had cure rate 92% in Marcove's first series and a preoperative embolization that was performed in five patients with result of no recurrence, were mentioned in these series. However a relatively large number of patients is not studied yet 22). Lee et al. 19) reported that bone cement injection offers an adjuvant strategy that may enhance the efficacy of treatment for GCT when complete en bloc spondylectomy is difficult. There are reports that bisphosphonates showed potential therapeutic benefits 1,913). Balke et al. 1) mentioned that most inoperable sacral GCTs that had repeatedly recurred did not increase in size with no further recurrence was seen. Although the role of bisphosphonates for treatment of GCT is still unknown, the administration of bisphosphonates can be considered in complicated cases and metastasis. Recently, denosumab has been used as a chemotherapeutic agent. Denosumab, a human monoclonal antibody against RANK ligand can inhibit the osteoclast function. Thomas et al. 29) reported that 35 patients of GCT were treated with denosumab, 30 cases were effective for tumor control after 25 weeks. In Branstetter et al. 5) study, 17 out of 20 patients were examined at various stages of treatment to distinguish clinical benefits from denosumab such as improved functional status or reduced pain. Denosumab is expected to be a treatment option for complicated cases.
CONCLUSION
GCT in the spine is difficult to resect completely due to the special structure of the spine and the invasive nature of the tumor. In addition, its rarity contributes to the challenging nature of the treatment.
En bloc wide excision is a well known a treatment of choice in order to manage GCTs. However, en bloc wide excision of the GCT in the spine is not easy without damaging neurovascular structures. The authors performed gross total removal rather than en bloc wide excision and obtained results. In case of STR, RT was beneficial in delaying tumor recurrence. RT can be considered good preventive treatment modality for further recurrence.
References
1. Balke M, Campanacci L, Gebert C, Picci P, Gibbons M, Taylor R, et al : Bisphosphonate treatment of aggressive primary, recurrent and metastatic giant cell tumour of bone. BMC Cancer 2010, 10 : 462,     2. Balke M, Schremper L, Gebert C, Ahrens H, Streitbuerger A, Koehler G, et al : Giant cell tumor of bone : treatment and outcome of 214 cases. J Cancer Res Clin Oncol 2008, 134 : 969-978,   3. Bertoni F, Present D, Sudanese A, Baldini N, Bacchini P, Campanacci M : Giant-cell tumor of bone with pulmonary metastases. Six case reports and a review of the literature. Clin Orthop Relat Res 1988, (237):275-285,
4. Blackley HR, Wunder JS, Davis AM, White LM, Kandel R, Bell RS : Treatment of giant-cell tumors of long bones with curettage and bone-grafting. J Bone Joint Surg Am 1999, 81 : 811-820,   5. Branstetter DG, Nelson SD, Manivel JC, Blay JY, Chawla S, Thomas DM, et al : Denosumab induces tumor reduction and bone formation in patients with giant-cell tumor of bone. Clin Cancer Res 2012, 18 : 4415-4424,   6. Campanacci M, Baldini N, Boriani S, Sudanese A : Giant-cell tumor of bone. J Bone Joint Surg Am 1987, 69 : 106-114,   7. Campanacci M, Bertoni F, Bacchini P : Bone and Soft Tissue Tumors. Bologna : Springer, 1990
8. Carrasco CH, Murray JA : Giant cell tumors. Orthop Clin North Am 1989, 20 : 395-405,  9. Cheng YY, Huang L, Lee KM, Xu JK, Zheng MH, Kumta SM : Bisphosphonates induce apoptosis of stromal tumor cells in giant cell tumor of bone. Calcif Tissue Int 2004, 75 : 71-77,    10. Dahlin DC : Giant-cell tumor of vertebrae above the sacrum : a review of 31 cases. Cancer 1977, 39 : 1350-1356,   11. Feigenberg SJ, Marcus RB Jr, Zlotecki RA, Scarborough MT, Berrey BH, Enneking WF : Radiation therapy for giant cell tumors of bone. Clin Orthop Relat Res 2003, ( 411):207-216,   12. Fidler MW : Surgical treatment of giant cell tumours of the thoracic and lumbar spine : report of nine patients. Eur Spine J 2001, 10 : 69-77,   13. Gille O, Oliveira Bde A, Guerin P, Lepreux S, Richez C, Vital JM : Regression of giant cell tumor of the cervical spine with bisphosphonate as single therapy. Spine (Phila Pa 1976) 2012, 37 : E396-E399,   14. Goldenberg RR, Campbell CJ, Bonfiglio M : Giant-cell tumor of bone. An analysis of two hundred and eighteen cases. J Bone Joint Surg Am 1970, 52 : 619-664,   15. Hart RA, Boriani S, Biagini R, Currier B, Weinstein JN : A system for surgical staging and management of spine tumors. A clinical outcome study of giant cell tumors of the spine. Spine (Phila Pa 1976) 1997, 22 : 1773-1782, discussion 1783   16. Kim S, Kim Y, You S, Kim S, Park I, Baik M : The trend of treatment for giant cell tumors of the spine in recent years. Age 2003, 30 : 68,
17. Lackman RD, Khoury LD, Esmail A, Donthineni-Rao R : The treatment of sacral giant-cell tumours by serial arterial embolisation. J Bone Joint Surg Br 2002, 84 : 873-877,   18. Lausten GS, Jensen PK, Schi├Ėdt T, Lund B : Local recurrences in giant cell tumour of bone. Long-term follow up of 31 cases. Int Orthop 1996, 20 : 172-176,   19. Lee CG, Kim SH, Kim DM, Kim SW : Giant cell tumor of upper thoracic spine. J Korean Neurosurg Soc 2014, 55 : 167-169,    20. Liljenqvist U, Lerner T, Halm H, Buerger H, Gosheger G, Winkelmann W : En bloc spondylectomy in malignant tumors of the spine. Eur Spine J 2008, 17 : 600-609,    21. Malek F, Krueger P, Hatmi ZN, Malayeri AA, Faezipour H, O'Donnell RJ : Local control of long bone giant cell tumour using curettage, burring and bone grafting without adjuvant therapy. Int Orthop 2006, 30 : 495-498,    22. Marcove RC, Lyden JP, Huvos AG, Bullough PB : Giant-cell tumors treated by cryosurgery. A report of twenty-five cases. J Bone Joint Surg Am 1973, 55 : 1633-1644,   23. Meyers SP, Yaw K, Devaney K : Giant cell tumor of the thoracic spine : MR appearance. AJNR Am J Neuroradiol 1994, 15 : 962-964,   24. Mirra JM, Gold RH, Marcove RC : Bone Tumors, Diagnosis and Treatment : Diagnosis and Treatment. Philadelphia : Lippincott Williams & Wilkins, 1980
25. Ozaki T, Liljenqvist U, Halm H, Hillmann A, Gosheger G, Winkelmann W : Giant cell tumor of the spine. Clin Orthop Relat Res 2002, ( 401):194-201,   26. Salzer-Kuntschik M : [Differential diagnosis of giant cell tumor of bone]. Verh Dtsch Ges Pathol 1998, 82 : 154-159,  27. Sanjay BK, Sim FH, Unni KK, McLeod RA, Klassen RA : Giant-cell tumours of the spine. J Bone Joint Surg Br 1993, 75 : 148-154,   28. Savini R, Gherlinzoni F, Morandi M, Neff JR, Picci P : Surgical treatment of giant-cell tumor of the spine. The experience at the Istituto Ortopedico Rizzoli. J Bone Joint Surg Am 1983, 65 : 1283-1289,   29. Thomas D, Henshaw R, Skubitz K, Chawla S, Staddon A, Blay JY, et al : Denosumab in patients with giant-cell tumour of bone : an open-label, phase 2 study. Lancet Oncol 2010, 11 : 275-280,  
Fig.┬Ā1
Recurrence free period of patients with giant cell tumor. Median value of recurrence free period is 84 months.
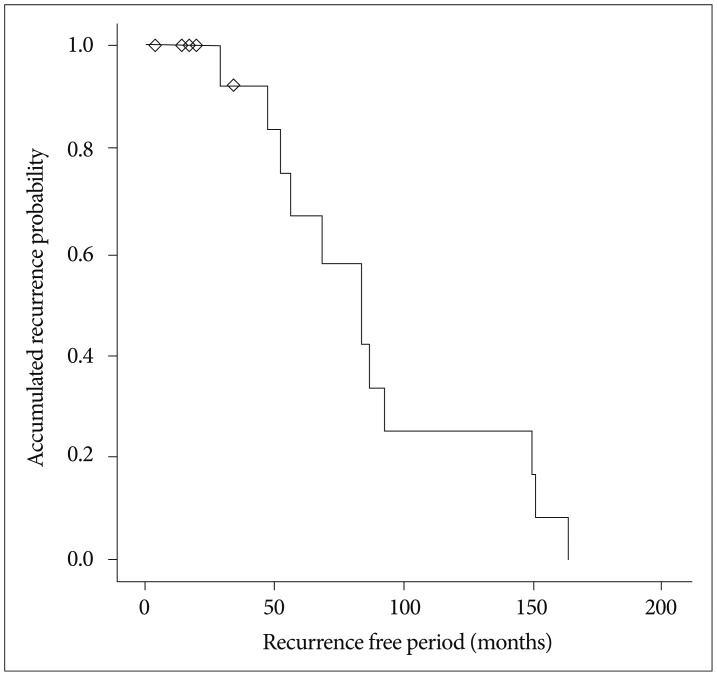
Fig.┬Ā2
Average value of recurrecnce free period is 112 months in radiation therapy group and 65 months in non-radiation therapy group (p=0.041).
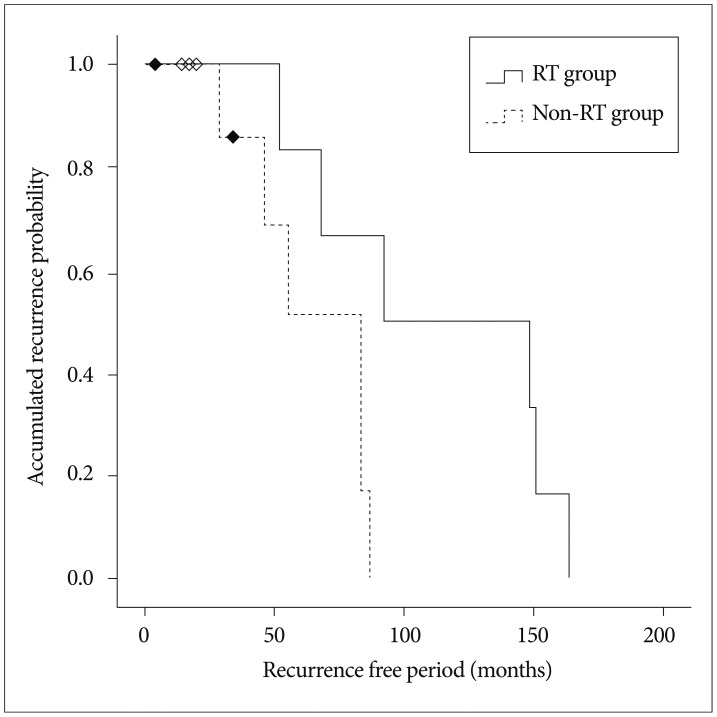
Fig.┬Ā3
A : Preoperative magnetic resonance (MR) image. Heterogeneously enhancing huge mass showed in sacrum and presacral area. Involvement of sacral foramen and right sacroiliac joint. B : Postoperative computed tomography scan. Remained bony destruction of sacrum with low attenuating and globular enhancing lesion. C : MR image when recurrence. Increased extent of heterogeneously enhancing lesion in remained sacrum and presacral mass. Enhancing mass progressed to the left side in paraspinal muscle and sacroiliac joint. D : Last follow-up MR image. No displaying signs of recurrence 94 months after the reoperation.
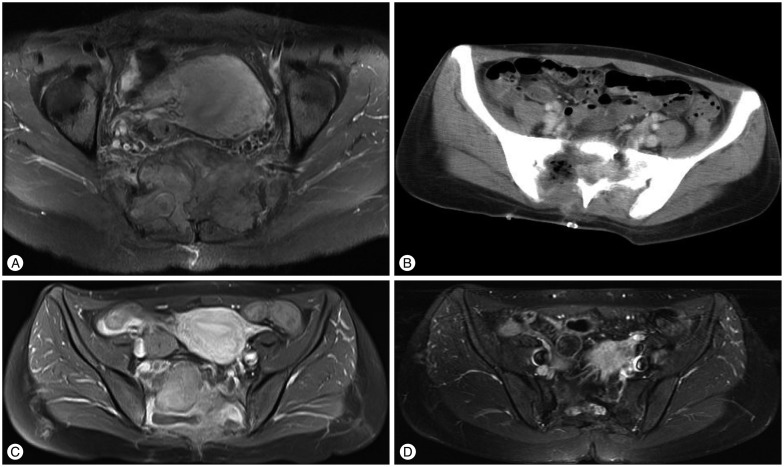
Fig.┬Ā4
A : Preoperative MR image. Heterogeneously enhancing mass involving in sacrum, sacral canal, right sacroiliac joint and pelvic cavity. B : Postoperative MR image. Remaining enhancing lesion in sacrum and right sacroiliac joint. C : Cyberknife® radiosurgery (marginal dose 3000 cGy) was performed. D : Last follow-up MR image. No change in enhancing lesion for 68 months.
Table┬Ā1
Demographic data of 19 patients with giant cell tumors
Table┬Ā2
Recurrence of giant cell tumor

|
|





















