INTRODUCTION
Recently developed motion-preserving spinal implants that consist of new synthetic materials, including disc replacements and dynamic posterior stabilization systems, may cause a foreign body reaction (FBR) around the implant itself or due to unintended wear debris. Wear and tear on the implant may occur by repetitive spinal motion in daily life, which can induce a FBR against the wear particles. When this condition becomes chronic, local osseous and epidural soft-tissue structures can be affected4). In addition, a FBR and fibrous encapsulation develop around the implant even in the absence of wear particles by a series of inflammatory and wound healing processes2). Here, we present a case of FBR causing loosening of a device for intervertebral assisted motion (DIAM; Medtronic Spine and Biologics, Memphis, TN, USA) 5 years after implantation. To the best of the author's knowledge, a FBR due to polyethylene wear after implantation of a DIAM has been reported only once previously7).
CASE REPORT
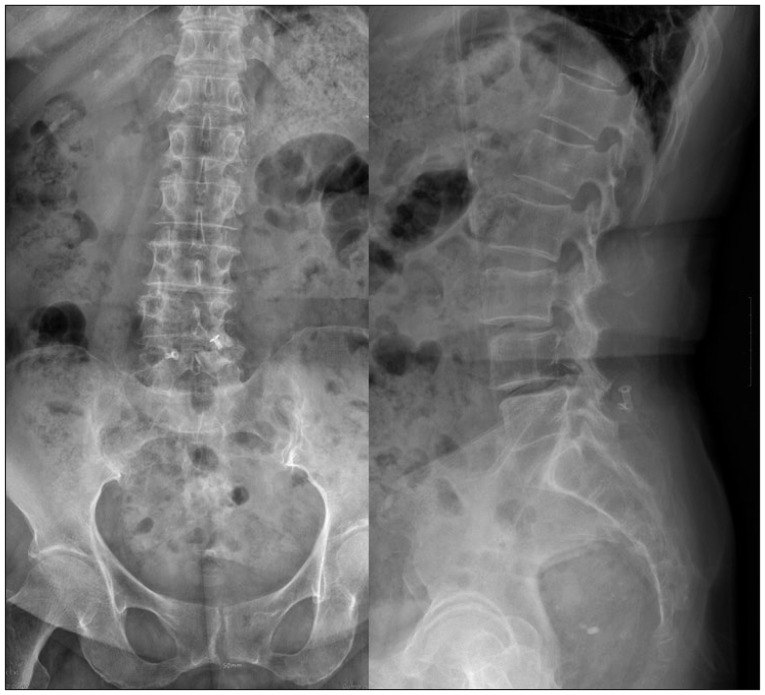
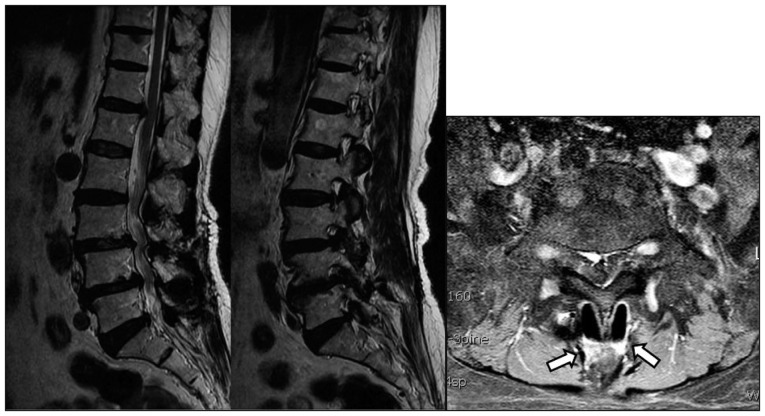
A 72-year-old female visited our hospital for intractable lower back pain that had radiated into her legs for 1 month. She had undergone decompressive laminotomy and DIAM insertion at L4-5 for spinal stenosis 5 years previously. Two years later, she underwent decompressive laminotomy at L3-4 for newly developed spinal stenosis at the adjacent segments. Plain lateral radiographs showed degenerative spondylolisthesis of L3-4 and collapse of the intervertebral disc space of L4-5, as compared to the initial plain radiographs. Plain anteroposterior radiographs showed scoliotic changes due to left sided collapse of the L4-5 disc space, which were not seen on immediate postoperative radiographs (Fig. 1). MRI showed L3-4 thecal sac compression and left L4-5 foraminal stenosis. High signal changes indicating fluid collection around the implanted DIAM were also seen (Fig. 2). Laboratory tests revealed an elevated erythrocyte sedimentation rate of 65 mm/h but a normal C-reactive protein level of 0.08 mg/dL. The white blood cell count was 5.05√Ч109/L.
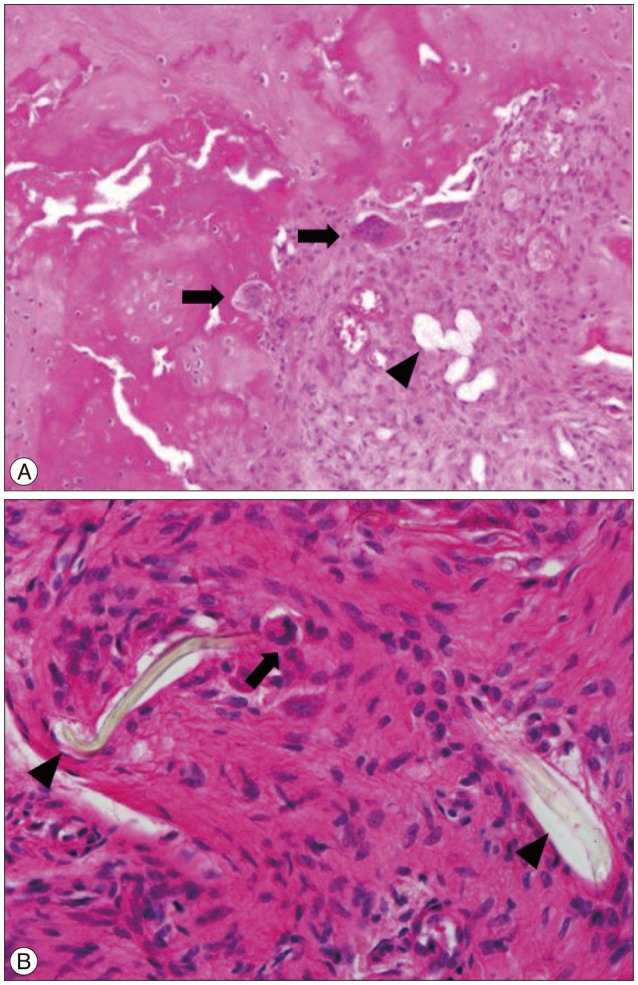
The patient underwent removal of the DIAM system and instrumented fusion at L3-5. Surgical exploration revealed that the DIAM had loosened around the inflammatory granulation tissues and was surrounded by scant serous fluid. The soft tissues around the DIAM were sampled for pathology. Histopathology showed numerous wear particles scattered with chronic inflammatory cells and foreign body giant cells (FBGCs) (Fig. 3). Bacterial cultures of the biopsies were negative.
Her back pain improved after the L3-5 fusion surgery. The visual analogue scale score of her back pain improved from 10 preoperatively to 4 at 2 weeks postoperatively and to 1 at 2 years postoperatively.
DISCUSSION
The DIAM is an interspinous distraction device that consists of an X-shaped silicone (dimethylsiloxane) interspinous process bumper enveloped by a polyethylene terephthalate (PET) fiber sack8). It acts as a dynamic stabilizer that preserves motion and provides segmental stabilization. However, Ha et al.6) reported that the DIAM had lost its indirect decompression by distraction that may be caused by loss of the elasticity of the components of the implant.
The allowed spinal motion paradoxically might stress the implant itself. Wear and tear can develop and eventually lead to long-term issues in plastic surgery, such as silicone granulomas9). In this process, the PET or silicone that the DIAM is made from can produce wear debris that then leads to a FBR. In addition, foreign bodies can be introduced during the surgical procedure itself. The microarchitecture of the PET fiber sac can also be damaged, as one wing of the implant is squeezed and compressed during insertion between the interspinous processes.
Cunningham et al.4) reported that during the epidural application of PET and silicone, the debris can elicit a chronic histiocytic reaction. In addition, an FBR can develop around the material itself, even in the absence of wear debris10). Macrophages and FBGCs are critical in the formation of an FBR3). In our case, fibrils from the PET fiber sac were scattered in the tissue around the DIAM and acted as foreign bodies. Histiocytes, polymorphous inflammatory cells, and FBGCs had infiltrated the space around the implant. Polyethylene debris, as in total joint replacement, is taken up by macrophage giant cells that release prostaglandin E2, which resorbs bone, causing the implant to loosen and leading to a vicious cycle of wear and loosening5).
The lumbar spine is subject to much external force. It supports the body weight and is a highly mobile region. An implanted DIAM also has to endure the stress of body weight and lumbar motion. Even if most implants induce FBR in some degree, lumbar motion and body weight can jeopardize the interspinous device, accelerating its wear and tear. The spinous process around the DIAM can be resorbed and the implant can loosen, eventually causing lower back pain and/or radiating pain1). In this case, the back and radiating pain were assumed to be caused by implant loosening and segmental instability. Disc space collapse in both the coronal and sagittal planes might give rise to foraminal stenosis. Although we were unable to differentiate the origin of the lower back pain as from post-laminectomy instability or FBR, macrophages and FBGCs were demonstrated on histological examination. The patient improved markedly after removing most of the granulation tissue, profuse irrigation, and L3-4 fusion. We believe that the FBR contributed markedly to her symptoms preoperatively.
CONCLUSION
A FBR from wear debris of a DIAM can induce a hypersensitivity reaction and bone resorption around the implant, causing it to loosen. Therefore, surgeons should insert the DIAM meticulously so as not to generate wear debris during the surgery. In patients with a DIAM who complain of back and radiating pain, a FBR must be considered, and the patients should be monitored long term.