INTRODUCTION
A spinal subdural hemorrhage (SDH) is a rare clinical entity that accounts for only 4.1% of all spinal hematomas13). The most common form of intra-spinal hemorrhage occurs in the epidural space26), and the lower incidence of SDH in the spine has been attributed to the protection of the spinal subdural space by the vertebrae, broad paravertebral muscles, and the rare passage of blood vessels such as bridging veins through the subdural space11). Spinal SDH is most frequently observed around the thoracic and thoracolumbar regions12).
The reasons for development of spinal SDH include hemorrhagic disorders, traumas, spinal surgery, lumbar puncture, or spinal anesthesia1,5). In addition, spinal vascular malformations or spinal tumors are sometimes reported as the causes of spinal SDH7). Diabetes, chronic kidney failure, and alcoholism are among the other etiological reasons.
Although the incidence of spinal SDH is quite low, it can also occur without any risk factors. This kind of spinal SDH is called "spontaneous spinal subdural hematoma"5,7). Spontaneous spinal hematomas were first described more than 60 years ago, by Schiller et al.23). Most cases are frequently associated with underlying coagulopathy and those receiving anticoagulants or antiplatelet agents8).
Simultaneous cranial and spinal SDH has been reported in only a few cases11). To our knowledge, only one case of simultaneous cranial subarachnoid hemorrhage (SAH) and spinal SDH exists in the literature17). In this report, we discuss a female patient on anticoagulant after aortic valve replacement surgery who showed simultaneous spinal SDH and cranial SAH.
CASE REPORT
The 53-year-old female patient was transferred to the emergency room of our institute due to the sudden onset of headache, nausea, and vomiting, all of which developed while she was taking a shower. A neurological examination revealed a slightly drowsy conscious mentality, nuchal rigidity with bilateral weakness of the lower limbs (Grade I) and sensory loss below T4 dermatome. Anal tone and perineal sensory capability were also severely impaired. The patient complained of the initial sudden onset of a headache and giddiness followed by the acute onset of weakness in the lower limbs with backache.
The patient had undertaken aortic valve replacement surgery about 10 years ago, and had been taking oral anticoagulant (Coumadine 5 mg, daily) since the operation. Her blood chemistry showed a platelet count of 199├Ś103/┬ĄL, a prothrombin time of 46.5 s (normal range, 9.4-12.5 s), a partial thromboplastin time of 47.3 s (normal range, 28.0-44.0 s), and an International Normalized Ratio (INR) of 4.39 (range 0.9-1.27).
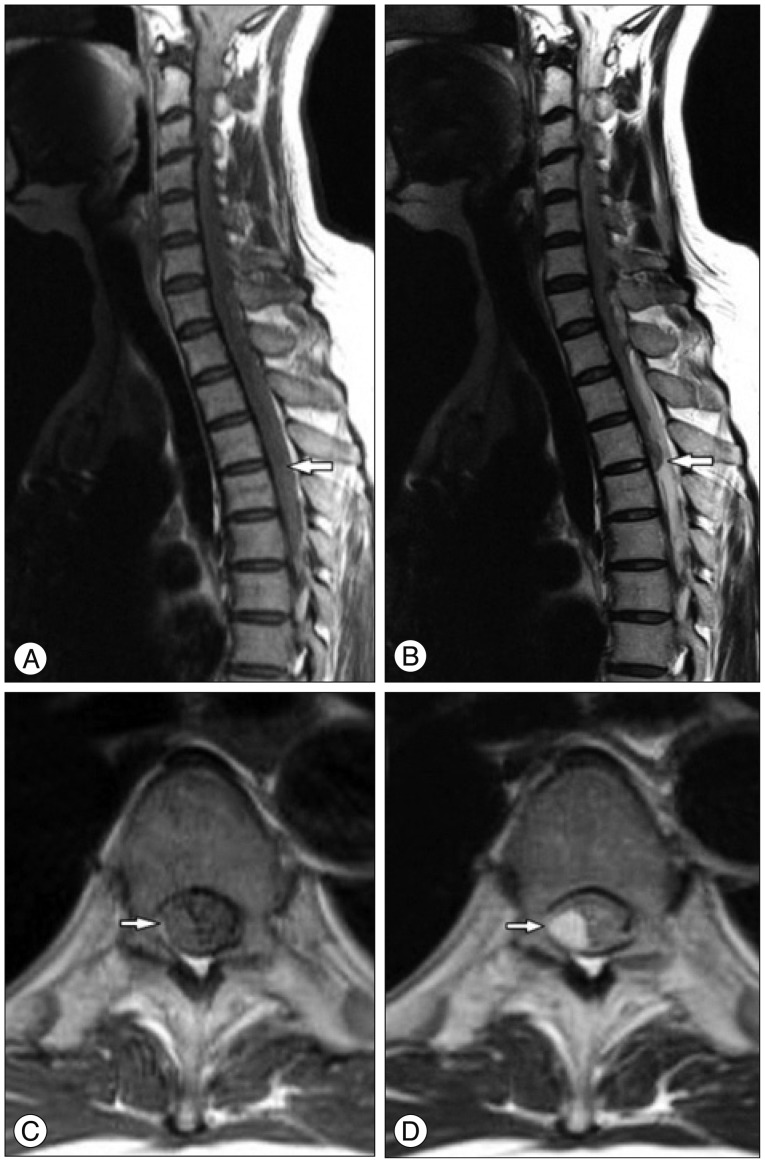
The patient underwent a brain computed tomography (CT) scan for assessing the impairment of her consciousness, which showed a SAH that extended symmetrically to the bilateral Sylvian and inter-hemispheric fissures with 3rd and 4th intraventricular hemorrhage (IVH) (Fig. 1). Under the impression of a ruptured anterior communicating cerebral artery aneurysm, an emergency trans-femoral cerebral angiography (TFCA) was performed to identify the exact location of the cerebral aneurysm following the brain CT scan. However, there was no aneurysmal sac or dissecting lesion on the cerebral angiography, so we decided to perform a whole spine magnetic resonance imaging (MRI) with contrast to check for the reason for the weakness of the bilateral lower limbs. The MRI revealed an acute subdural hematoma in the dorsal portion dominantly extending from C7 to T6 in the spinal canal causing a posterior compression of the spinal cord (Fig. 2).
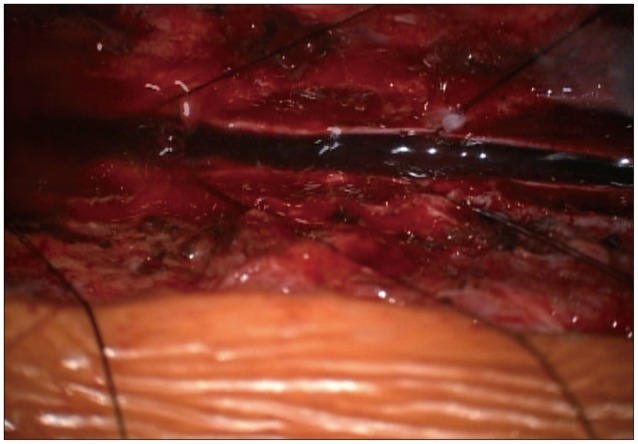
Vitamin K was administered to get the INR below two, and the patient underwent an emergency operation with planning for an extensive multilevel laminectomy and the removal of hematoma to release the pressure off the spinal cord. Under a laminectomy on C7-T6, the dura was opened longitudinally under a microscope, which revealed a dark-colored blood clot underneath (Fig. 3). The blood clot was then removed using blunt dissection and cleared with an intermittent irrigation and suction method to release the spinal cord from compression.
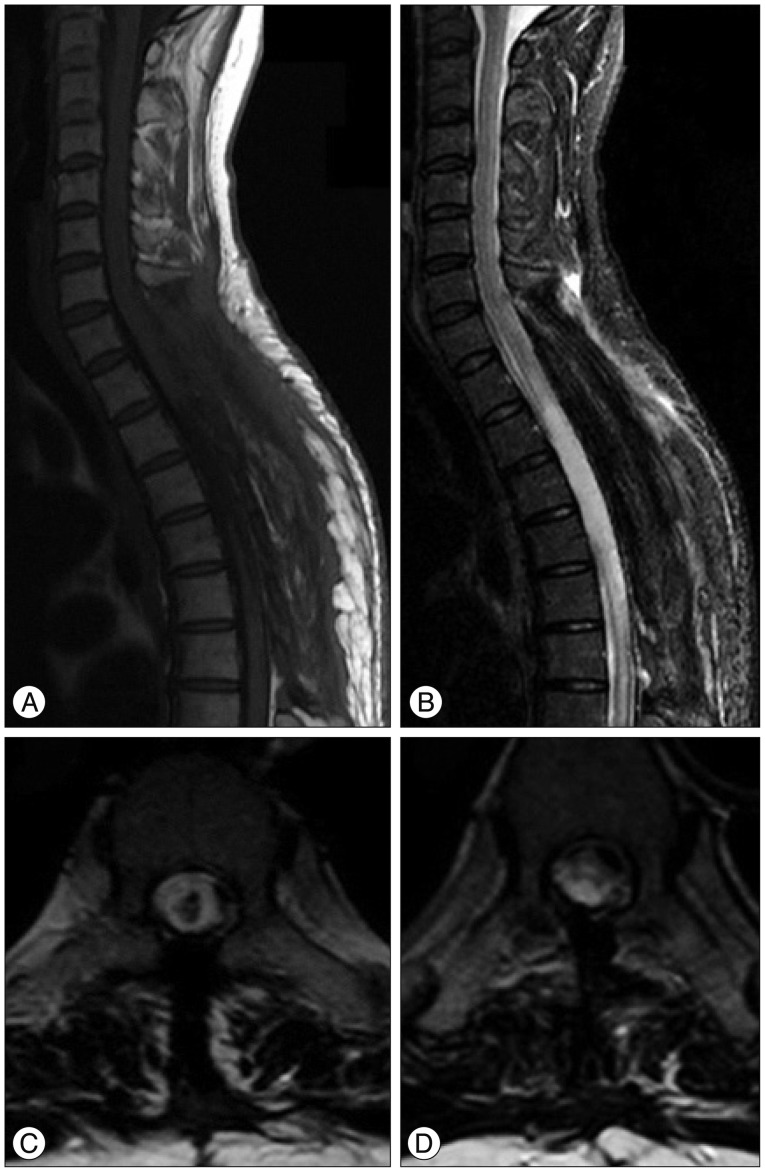
The patient presented no vasospasm, hydrocephalus, or any improvement of neurological deficit on either lower limb after the decompression surgery. Two weeks later, she was transferred to a rehabilitation facility. Despite three months of additional rehabilitation, there was no dramatic improvement in neurological impairment including the lower limbs paraparesis and sensory deficit apart from a slight improvement of anal tone and bladder function allowing a clean intermittent catheterization for voiding. The female patient has been complaining of neuropathic pain in both lower extremities, and the follow-up MRI shows malatic changes of the spinal cord where the hemorrhage occurred (8 months postoperatively) (Fig. 4).
DISCUSSION
Spinal SDH most frequently occurs in the thoracic spine. It manifests itself in sudden back pain that radiates to the upper or lower extremities or to the trunk and creates motor, sensory, and autonomic disturbances to a variable degree14). Most cases are seen in adults above 50 years old, with roughly half of the patients having predisposing factors such as coagulopathy or a history of spinal tap16). In particular, anticoagulated patients undergoing subdural or epidural anesthesia are at an increased risk of developing hematomas in the epidural or subdural space around the spinal cord4). Lieberman et al.15) reported a significant hemorrhage in 9.4% of patients treated with anticoagulants during an eight-year study period. The overall frequency of fatal hemorrhaging is 1.0%.
Unlike the intracranial subdural space, the spinal subdural space is an avascular6), which means that a hemorrhage in a subdural space might come from a subarachnoid source. The finding of an accompanying SAH in several cases with spinal SDH supports this hypothesis. A hemorrhage from the subarachnoid space extends to the subdural space when the arachnoid membrane ruptures. In addition, isolated spinal SDH may remain (usually in a ventral portion) after a more rapid resolution of a SAH by dilution with CSF14,16).
The postulated mechanism that causes non-traumatic and spontaneous spinal SDH is a sudden increase in the pressure of the intra-spinal vessels due to the indirect reflection of intra-abdominal or intra-thoracic pressure. Secondary to the increased intra-thoracic pressure because of a minor spinal trauma or sudden physical activity, the inner lumen pressure is increased in the venous structures located in the subarachnoid area. In the meantime, when the pressure of cerebrospinal fluid drops below the intravascular pressure values, it results in the laceration of the venous structures such as the valveless radiculomedullary veins, which cross the spinal subarachnoid space17,18).
A contradictory hypothesis exists, however. Morandi et al.18) emphasized that the thin and delicate extra-arachnoid veins in the inner surface of the dura would lead to a subdural hematoma. Hung et al.9) hypothesized that a rise in the intracranial pressure could also increase the shearing force between the spinal subarachnoid and the subdural spaces, causing the inner dura to tear and bleed. Moreover, Schwartz et al.24), described the acute spinal SDH as being extra-arachnoidal, adding that it appeared to be between two layers of dura, suggesting that a potential intra-dural space could be the source of the bleeding.
It is not clear which is the main mechanism in the simultaneous development of cranial SAH and spinal SDH, and there are three possible theories. The first is that the cranial SAH and spinal SDH occurred simultaneously in different sites, as an extremely rare incidence. The second is that the cranial SAH extended into the spinal subarachnoid space and then the spinal subdural space by exceeding or lacerating the arachnoid membrane. The last is a reverse form of the second possible mechanism in which the extension moves from the spinal to the cranial portion.
Barnett2) have noted that SAH comes from the rupture of a berry aneurysm in the Willis circle and may extend into the subdural space in the cranium. The authors state that it may result in a subdural hematoma around the spinal cord. Raised intracranial pressure might displace the hematoma directly into the skull base and spinal canal through a subdural space11,28). The anatomic continuity between the intracranial and spinal subdural spaces can be observed, and a simultaneous cranial SAH and spinal SDH could explain this phenomenon19,27). However, when we applied the above mechanism to our patient to explain the simultaneous intracranial and spinal hemorrhage, it was found to be insufficient. There was no cranial SDH, only an IVH to explain the development of the spinal SDH from the mechanism of hemorrhage migration from the cranial to the spinal portion. In addition, if the cranial SAH developed as a non-aneurysmal type, there would not be sufficient pressure to move the hematoma to the spinal canal and make an intracranial SDH. In addition, the extension of a large amount of a SAH from the cranium to the spinal portion could compress the spinal cord or lacerate the arachnoid membrane, resulting in the formation of a spinal SDH, which is also uncommon, even in a case of an aneurysmal SAH.
The initial treatment for spinal SDH should consist of a wide laminectomy and the evacuation of the hematoma. However, there are some cases in the literature that have been treated via conservative treatment, without any surgical intervention10,1124). The exact mechanism of the spontaneous resolution of the symptoms is unclear. The rapid redistribution of a hematoma with rostro-caudal expansion within a subdural space may avoid transverse expansions, and subsequently reduce the degree of cord compression20). Another possible mechanism was recently suggested, stating that the redistribution of the spinal SDH could occur due to the rapid dilution of the hematoma, intermingled with cerebrospinal fluid, in the presence of arachnoid membrane tearing16).
Intra-spinal hematoma is the first diagnosis that should be considered in patients on anticoagulant therapies, presenting itself as acute cord compression or cauda equina syndrome18,21). It is therefore advisable to assess the patients on anticoagulation therapy thoroughly for the possibility of an intra-spinal hematoma in order to avoid any major neurological complications. The spontaneous onset of spinal SDH in patients on an anticoagulation therapy is a neurological emergency. With this in mind, early diagnosis, the discontinuation of the anticoagulant, and urgent surgical decompression are recommended to allow for neurological recovery18,22).
The timing of the surgery and the anatomic location of the hematoma determine a patient's functional outcomes3,18). Additionally, the degree of preoperative neurological deficit and level of the SDH correlate significantly with the early and long-term functional outcomes, despite prompt evacuation18,25).
CONCLUSION
When a patient is taking anticoagulation therapy and presents with a subarachnoid hemorrhage without hydrocephalus and severe lower extremity paraparesis or paraplegia with backache, the possibility of spinal hemorrhage should be considered. The spontaneous development of a spinal SDH is a neurological emergency. Therefore, early diagnosis, the discontinuation of anticoagulants, and urgent surgical decompression are recommended to facilitate neurological recovery.